JEE Thermodynamics PYQs
The JEE Physics syllabus covers JEE Thermodynamics PYQs. They assist you in knowing the type of questions that are to be asked in this chapter and the familiarity of the key concepts, including, heat, temperature, work, internal energy, the first law of thermodynamics, specific heat, heat capacity, and thermodynamic processes.
Thermodynamics questions in the exam are normally in the form of direct numerical question or concept based questions. The positive thing is that this chapter is a lot easier when you are clear on your basics. When you get thoroughly familiar with the concepts and are aware of the formula to apply, it becomes a lot easier to resolve questions. There is no reason to consider thermodynamics a highly complicated chapter. It is one of the more scoring parts of JEE Physics, though with frequent revision and intelligent practice it can become that.
In this blog, you will find a simple formula PDF, a section for important JEE Thermodynamics PYQs in download format, a few practice questions with answers, and some extra questions to solve on your own. You will also learn about common mistakes students often make and a few simple tips to save time in the exam.
JEE Thermodynamics Important PYQs PDF
This PDF can include the most important previous year questions from thermodynamics. It may cover topics like heat, work, internal energy, specific heat, heat capacity, the first law of thermodynamics, isothermal process, adiabatic process, isobaric process, and isochoric process.
Practicing these questions will help you understand the exam pattern better. It will also improve your speed, accuracy, and confidence before the exam.
Important Formulas for JEE Thermodynamics PYQs
You only need a few important formulas and ideas to solve most thermodynamics questions in JEE. These formulas help you understand heat transfer, work done, energy change, and different thermodynamic processes more clearly.
You can download the full formula PDF from the link above. Here is a quick look at some of the main formulas:
Concept | Formula |
First Law of Thermodynamics | ΔQ = ΔU + ΔW |
Work Done by Gas | W = PΔV |
Heat Supplied | Q = mcΔT |
Heat Capacity | C = Q/ΔT |
Specific Heat Capacity | c = Q/mΔT |
Internal Energy of Ideal Gas | U = nCᵥT |
Isothermal Process | PV = constant |
Adiabatic Process | PVᵞ = constant |
Relation Between Cp and Cv | Cp − Cv = R |
Efficiency of Heat Engine | η = W/Q₁ |
These formulas are commonly used in questions based on heat, work, internal energy, heat capacity, and thermodynamic processes. If you revise them properly, many JEE questions start to feel much easier.
Top 5 Common Mistakes to Avoid in JEE Thermodynamics PYQs
Many students find thermodynamics confusing at first because it includes both concepts and formulas. But most mistakes happen because small details are missed while solving. Here are some common mistakes you should avoid:
Mixing up heat and temperature
Heat and temperature are related, but they are not the same. Heat is energy transfer, while temperature tells you how hot or cold a body is.
Using the wrong sign convention
In thermodynamics, signs matter a lot. Heat supplied to the system, work done by the system, and work done on the system must be handled carefully.
Confusing different thermodynamic processes
Isothermal, adiabatic, isobaric, and isochoric processes all follow different conditions. Many students mix them up and use the wrong formula.
Forgetting the first law of thermodynamics
Some students try to solve questions directly without checking how heat, work, and internal energy are connected. In many cases, the first law gives the easiest way to solve the problem.
Ignoring units
Always use proper SI units like joule, kelvin, mole, and pascal. Even if the method is correct, a unit mistake can change the final answer.
List of JEE Thermodynamics PYQs
Here is a short set of JEE-style thermodynamics questions for practice. These include common question types from heat, work, internal energy, and thermodynamic processes. Solving them regularly can help you become faster and more confident.
Question 1
A diatomic gas ( y= 1.4) does 100 J of work when it is expanded isobarically. Then the heat given to the gas ____ J.
correct answer:- 350
Question 2
10 mole of oxygen is heated at constant volume from $$30^{\circ}C \text{to} 40^{\circ}C$$. The change in the internal energy of the gas is ____ cal (the molecular specific heat of oxygen at constant pressure, $$C_{p}= 7 \text{cal}/\text{mol}.^{\circ}C \text{and} R = 2 \text{cal}./\text{mol}.^{\circ}C).$$
correct answer:- 500
Question 3
The volume of an ideal gas increases 8 times and temperature becomes $$(1/4)^{th}$$ of initial temperature during a reversible change. If there is no exchange of heat in this process $$(\triangle Q = 0)$$ then identify the gas from the following options (Assuming the gases given in the options are ideal gases):
correct answer:- 2
Question 4
10 mole of an ideal gas is undergoing the process showu in the figure. The heat involved in the process from $$P_{1}$$ to $$P_{2}$$ is $$\alpha$$ Joule(P_{1}= 21.7Pa and $$P_{2} = 30$$ Pa, $$C_{v}=21J/K.mol, R=8.3 J/mol.K.$$) The value od $$\alpha$$ is _________.
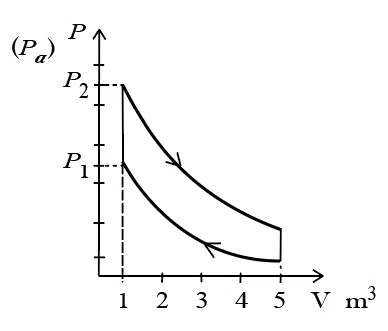
correct answer:- 4
Question 5
One mole of an ideal diatomic gas expands from volume $$V$$ to $$2 V$$ isothermally at a temperature $$27^{o}C$$ and does W joule of work. lf the gas undergoes same magnitude of expansion adiabatically from $$27^{o}C$$ doing the same amount of work $$W$$, then its final temperature will be (close to) ____ $$^{\circ}C.$$
$$(\log_{e}2 = 0.693)$$
correct answer:- 4
Question 6
A thermodynamic system is taken through the cyclic process $$ABC$$ as shown in the figure. The total work done by the system during the cycle $$ABC$$ is ______ J.
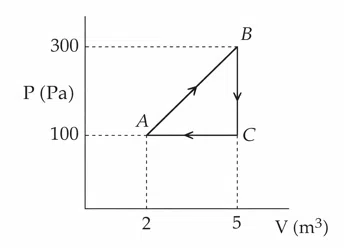
correct answer:- 300
Question 7
Given are statements for certain thermodynamic variables, (A) Internal energy, volume (V) and mass (M) are extensive variables. (B) Pressure (P), temperature (T) and density $$(\rho)$$ are intensive variables. (C) Volume (V), temperature (T) and density $$(\rho)$$ are intensive variables. (D) Mass (M), temperature (T) and internal energy are extensive variables. Choose the correct answer from the options given below :
correct answer:- 4
Question 8
Water of mass m gram is slowly heated to increase the temperature from $$T_{1}$$ to $$T_{2}$$ The change in entropy of the water, given specific heat of water is $$1Jkg^{-1}K^{-1}$$, is :
correct answer:- 1
Question 9
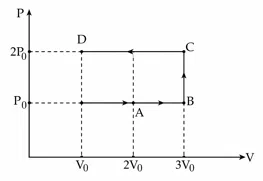
Using the given P - V diagram, the work done by an ideal gas along the path ABCD is :
correct answer:- 3
Question 10
An ideal gas goes from an initial state to final state. During the process, the pressure of gas increases linearly with temperature. A. The work done by gas during the process is zero. B. The heat added to gas is different from change in its internal energy. C. The volume of the gas is increased. D. The internal energy of the gas is increased. E. The process is isochoric (constant volume process) Choose the correct answer from the options given below:
correct answer:- 3
Question 11
The magnitude of heat exchanged by a system for the given cyclic process ABCA (as shown in figure) is (in SI unit) :
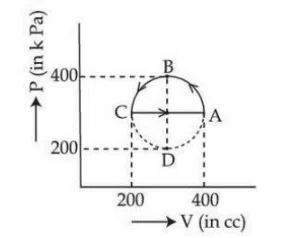
correct answer:- 1
Question 12
Given below are two statements. One is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : In an insulated container, a gas is adiabatically shrunk to half of its initial volume. The temperature of the gas decreases. Reason (R): Free expansion of an ideal gas is an irreversible and an adiabatic process. In the light of the above statements, choose the correct answer from the options given below :
correct answer:- 2
Question 13
A Carnot engine (E) is working between two temperatures 473 K and 273 K . In a new system two engines - engine $$E_{1}$$ works between 473 K to 373 K and engine $$E_{2}$$ works between 373 K to 273 K . If $$\eta_{12},\eta_{1}$$ and $$\eta_{2}$$ are the efficiencies of the engines $$E,E_{1}$$ and $$E_{2}$$, respectively, then
correct answer:- 4
Question 14
The workdone in an adiabatic change in an ideal gas depends upon only :
correct answer:- 1
Question 15

correct answer:- 1
Question 16
An ideal gas initially at $$0^{\circ}$$C temperature, is compressed suddenly to one fourth of its volume. If the ratio of specific heat at constant pressure to that at constant volume is 3/2, the change in temperature due to the thermodynamic process is _____ K.
correct answer:- 273
Question 17
Given below are two statements. One is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : With the increase in the pressure of an ideal gas, the volume falls off more rapidly in an isothermal process in comparison to the adiabatic process.
Reason (R) : In isothermal process, PV = constant, while in adiabatic process $$PV^{\gamma}$$ = constant. Here $$\gamma$$ is the ratio of specific heats, P is the pressure and V is the volume of the ideal gas. In the light of the above statements, choose the correct answer from the options given below :
correct answer:- 1
Question 18
The difference of temperature in a material can convert heat energy into electrical energy. To harvest the heat energy, the material should have
correct answer:- 4
Question 19
A cup of coffee cools from $$90^{\circ}$$ to $$80^{\circ}$$ in t minutes when the room temperature is $$20^{\circ}$$. The time taken by the similar cup of coffee to cool from $$80^{\circ}$$ to $$60^{\circ}$$ at the same room temperature is :
correct answer:- 4
Question 20
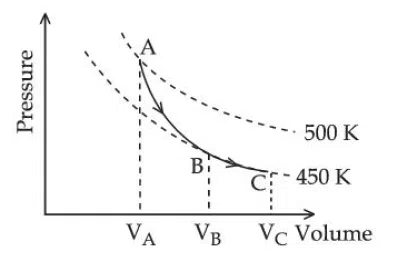
A poly-atomic molecule ($$C_V = 3R,\, C_P = 4R$$, where R is gas constant) goes from phase space point $$A\ (P_A = 10^5\,\text{Pa},\ V_A = 4 \times 10^{-6}\,\text{m}$$ to point $$B\ (P_B = 5 \times 10^4\,\text{Pa},\ V_B = 6 \times 10^{-6}\,\text{m}^3)$$ to point $$C\,(P_C = 10^4\,\text{Pa},\; V_C = 8 \times 10^{-6}\,\text{m}^3).$$ A to B is an adiabatic path B and C to is an isothermal path. The net heat absorbed per unit mole by the system is :
correct answer:- 2
Question 21
P-T diagram of an ideal gas having three different densities $$\rho_1, \rho_2, \rho_3$$ (in three different cases) is shown in the figure. Which of the following is correct:
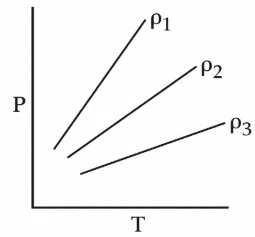
correct answer:- 1
Question 22
The pressure and volume of an ideal gas are related as $$PV^{3/2} = K$$ (Constant). The work done when the gas is taken from state $$A(P_1, V_1, T_1)$$ to state $$B(P_2, V_2, T_2)$$ is :
correct answer:- 1
Question 23
A diatomic gas $$(\gamma = 1.4)$$ does 200 J of work when it is expanded isobarically. The heat given to the gas in the process is:
correct answer:- 4
Question 24
$$0.08$$ kg air is heated at constant volume through $$5°C$$. The specific heat of air at constant volume is $$0.17 \text{ kcal kg}^{-1} \text{ °C}^{-1}$$ and $$1 \text{ J} = 4.18 \text{ joule cal}^{-1}$$. The change in its internal energy is approximately.
correct answer:- 3
Question 25
During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its absolute temperature. The ratio of $$\frac{C_p}{C_v}$$ for the gas is :
correct answer:- 2
Question 26
A thermodynamic system is taken from an original state $$A$$ to an intermediate state $$B$$ by a linear process as shown in the figure. Its volume is then reduced to the original value from $$B$$ to $$C$$ by an isobaric process. The total work done by the gas from $$A$$ to $$B$$ and $$B$$ to $$C$$ would be :
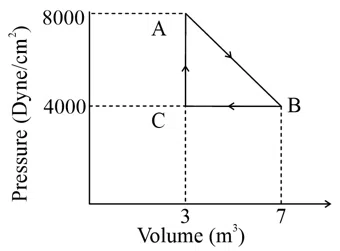
correct answer:- 4
Question 27
Two thermodynamical processes are shown in the figure. The molar heat capacity for process $$A$$ and $$B$$ are $$C_A$$ and $$C_B$$. The molar heat capacity at constant pressure and constant volume are represented by $$C_P$$ and $$C_V$$, respectively. Choose the correct statement.
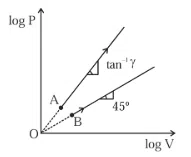
correct answer:- 1
Question 28
Choose the correct statement for processes $$A$$ & $$B$$ shown in figure.
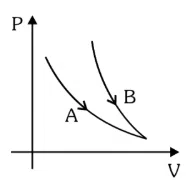
correct answer:- 1
Question 29
The given figure represents two isobaric processes for the same mass of an ideal gas, then
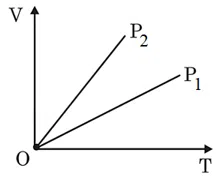
correct answer:- 4
Question 30
A sample of gas at temperature $$T$$ is adiabatically expanded to double its volume. Adiabatic constant for the gas is $$\gamma = 3/2$$. The work done by the gas in the process is: ($$\mu = 1$$ mole)
correct answer:- 3
Question 31
Match List-I with List-II :

Choose the correct answer from the options given below :
correct answer:- 2
Question 32
During an adiabatic process, if the pressure of a gas is found to be proportional to the cube of its absolute temperature, then the ratio of $$\frac{C_P}{C_V}$$ for the gas is :
correct answer:- 3
Question 33
The heat absorbed by a system in going through the given cyclic process is :
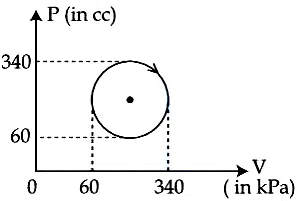
correct answer:- 2
Question 34
To project a body of mass $$m$$ from earth's surface to infinity, the required kinetic energy is (assume, the radius of earth is $$R_E$$, $$g =$$ acceleration due to gravity on the surface of earth):
correct answer:- 3
Question 35
The specific heat at constant pressure of a real gas obeying $$PV^2 = RT$$ equation is:
correct answer:- 3
Question 36
A total of $$48 \text{ J}$$ heat is given to one mole of helium kept in a cylinder. The temperature of helium increases by $$2°C$$. The work done by the gas is: Given, $$R = 8.3 \text{ J K}^{-1} \text{ mol}^{-1}$$.
correct answer:- 4
Question 37
Energy of 10 non rigid diatomic molecules at temperature $$T$$ is :
correct answer:- 2
Question 38
Two different adiabatic paths for the same gas intersect two isothermal curves as shown in P-V diagram. The relation between the ratio $$\frac{V_a}{V_d}$$ and the ratio $$\frac{V_b}{V_c}$$ is:
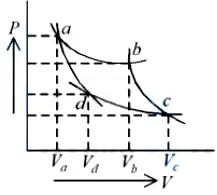
correct answer:- 2
Question 39
The volume of an ideal gas $$(\gamma = 1.5)$$ is changed adiabatically from 5 litres to 4 litres. The ratio of initial pressure to final pressure is:
correct answer:- 3
Question 40
A real gas within a closed chamber at $$27°C$$ undergoes the cyclic process as shown in figure. The gas obeys $$PV^3 = RT$$ equation for the path $$A$$ to $$B$$. The net work done in the complete cycle is (assuming $$R = 8 \text{ J/molK}$$):
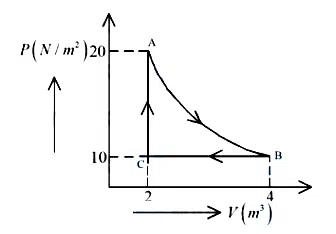
correct answer:- 2

 Group
Group






.webp)



















