Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The specific heat at constant pressure of a real gas obeying $$PV^2 = RT$$ equation is:
For a real gas, the relation between the molar specific heats is
$$C_P - C_V \;=\; T\Bigl(\frac{\partial P}{\partial T}\Bigr)_V \Bigl(\frac{\partial V}{\partial T}\Bigr)_P$$
This identity is obtained from the first & second laws together with exact-differential criteria, and it is valid for one mole of any substance.
The gas in the question obeys the equation of state
$$P\,V^2 \;=\; R\,T \;\;-(1)$$
Step 1: Find $$\Bigl(\dfrac{\partial P}{\partial T}\Bigr)_V$$.
From $$(1)$$: $$P \;=\; \dfrac{R\,T}{V^{2}}$$. Keeping $$V$$ constant and differentiating with respect to $$T$$:
$$\Bigl(\frac{\partial P}{\partial T}\Bigr)_V = \frac{R}{V^{2}} \;\;-(2)$$
Step 2: Find $$\Bigl(\dfrac{\partial V}{\partial T}\Bigr)_P$$.
Rewrite $$(1)$$ to express $$V$$ in terms of $$T$$ at fixed $$P$$:
$$V^{2} = \frac{R\,T}{P} \quad\Longrightarrow\quad 2V\,dV = \frac{R}{P}\,dT$$ so
$$\Bigl(\frac{\partial V}{\partial T}\Bigr)_P = \frac{R}{2\,P\,V} \;\;-(3)$$
Step 3: Compute $$C_P - C_V$$.
Substitute $$(2)$$ and $$(3)$$ into the heat-capacity relation:
$$\begin{aligned} C_P - C_V &= T\left(\frac{R}{V^{2}}\right)\left(\frac{R}{2\,P\,V}\right)\\[4pt] &= \frac{T\,R^{2}}{2\,P\,V^{3}} \;\;-(4) \end{aligned}$$
Step 4: Eliminate $$P$$ using the equation of state.
From $$(1)$$: $$P = \dfrac{R\,T}{V^{2}}$$. Insert this in $$(4)$$:
$$\begin{aligned} C_P - C_V &= \frac{T\,R^{2}}{2\,(\tfrac{R\,T}{V^{2}})\,V^{3}}\\[6pt] &= \frac{T\,R^{2}}{2\,R\,T\,V}\\[4pt] &= \frac{R}{2\,V} \end{aligned}$$
Therefore
$$C_P = C_V + \frac{R}{2\,V}$$
Among the given options this matches Option C.
Answer: Option C
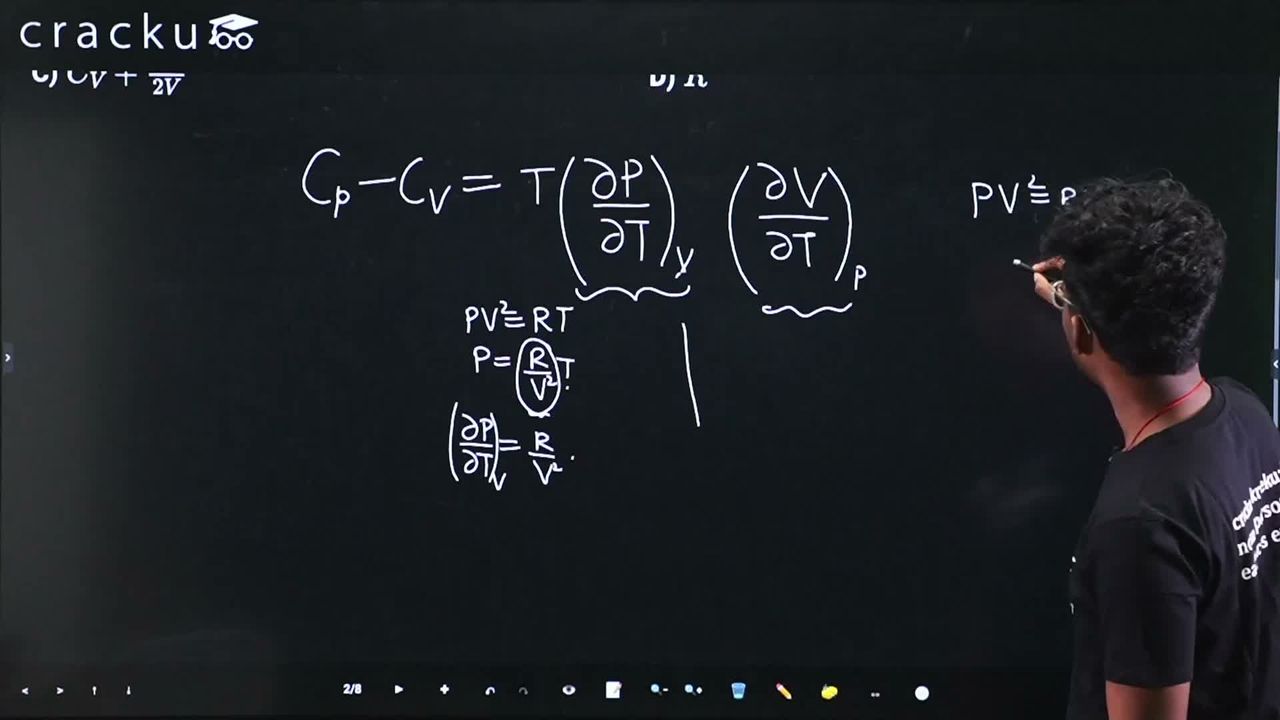
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
