Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
A sample contains mixture of helium and oxygen gas. The ratio of root mean square speed of helium and oxygen in the sample, is:
We need to find the ratio of r.m.s. speeds of helium to oxygen at the same temperature.
Recall the r.m.s. speed formula.
$$ v_{rms} = \sqrt{\frac{3RT}{M}} $$
where $$R$$ is the gas constant, $$T$$ is absolute temperature, and $$M$$ is the molar mass.
Calculate the ratio.
At the same temperature, $$R$$ and $$T$$ are the same for both gases:
$$ \frac{v_{He}}{v_{O_2}} = \sqrt{\frac{M_{O_2}}{M_{He}}} $$
Molar masses: $$M_{He} = 4$$ g/mol, $$M_{O_2} = 32$$ g/mol
$$ \frac{v_{He}}{v_{O_2}} = \sqrt{\frac{32}{4}} = \sqrt{8} = 2\sqrt{2} $$
The correct answer is Option (2): $$2\sqrt{2}$$.
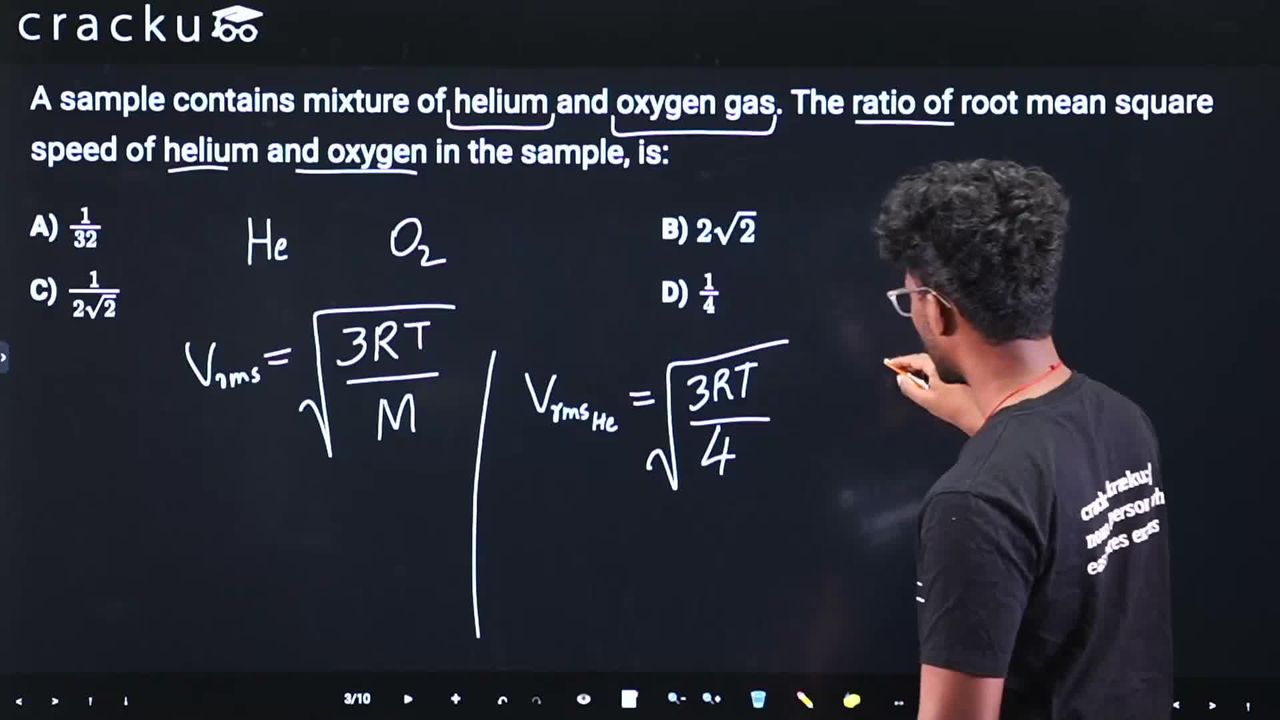
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
