Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
10 mole of an ideal gas is undergoing the process showu in the figure. The heat involved in the process from $$P_{1}$$ to $$P_{2}$$ is $$\alpha$$ Joule(P_{1}= 21.7Pa and $$P_{2} = 30$$ Pa, $$C_{v}=21J/K.mol, R=8.3 J/mol.K.$$) The value of $$\alpha$$ is _________.
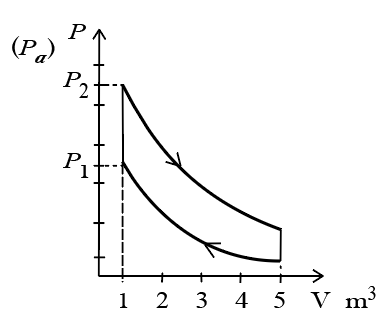
From the PV diagram, the two curved paths are adiabatic (their shape indicates $$PV^{\gamma}=\text{constant}$$, and the process from $$P_1$$ to $$P_2$$ at $$V=1m^3$$ is vertical, i.e. an isochoric process.
Since volume is constant, work done is zero:
W=0
So heat supplied equals change in internal energy:
$$Q=nC_v(T_2-T_1)$$
Using ideal gas law,
$$T_1=\frac{P_1V}{nR}=\frac{21.7\times1}{10\times8.3}$$
$$T_2=\frac{P_2V}{nR}=\frac{30\times1}{10\times8.3}$$
Thus,
$$T_2−T_1=\frac{\left(30−21.7\right)}{83}=\frac{8.3}{83}=0.1K$$
Therefore,
$$Q=10\times21\times0.1$$
$$Q=21J$$
So,
$$α=21$$
Create a FREE account and get:
Educational materials for JEE preparation
