Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Water of mass m gram is slowly heated to increase the temperature from $$T_{1}$$ to $$T_{2}$$ The change in entropy of the water, given specific heat of water is $$1Jkg^{-1}K^{-1}$$, is :
For reversible heating,
$$dS=\frac{dQ}{T}$$
For water,
$$dQ=mcdT$$
so
$$dS=\frac{mcdT}{T}$$
Integrating from $$T_1\ to\ T_2$$,
$$ΔS=\int^{_{ }}\frac{mcdT}{T}$$
$$\Delta S=mc\ln\left(\frac{T_2}{T_1}\right)$$
Given specific heat
$$c=1\ \text{J kg}^{-1}\text{K}^{-1}$$
Thus
$$\Delta S=m\ln\left(\frac{T_2}{T_1}\right)$$
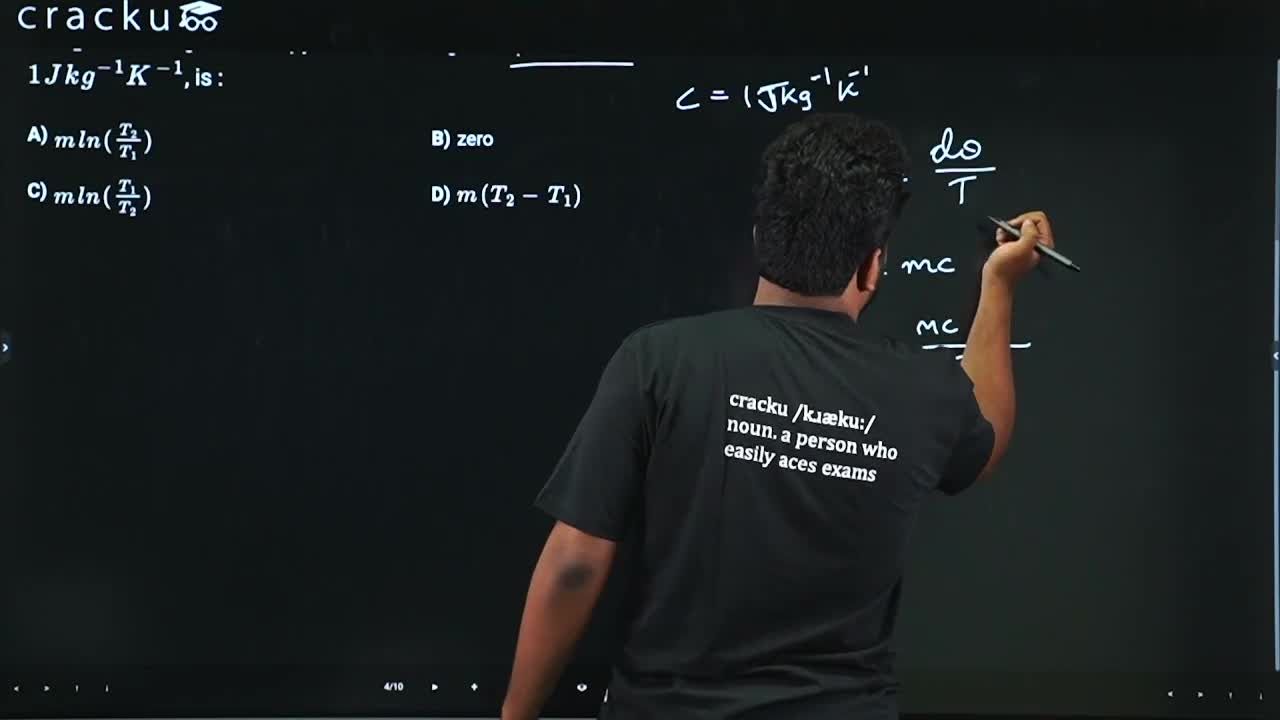
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
