Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
P-T diagram of an ideal gas having three different densities $$\rho_1, \rho_2, \rho_3$$ (in three different cases) is shown in the figure. Which of the following is correct:
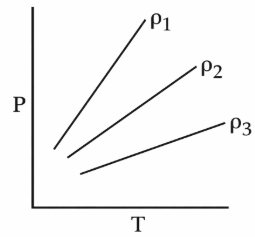
For an ideal gas,
$$PV=nRT$$
Also density,
$$\rho=\frac{m}{V}$$
and
$$n=\frac{m}{M}$$
So ideal gas law becomes
$$P=\frac{\rho}{M}RT$$
or
$$P=\left(\frac{\rho R}{M}\right)T$$
This is the equation of a straight line in a P-T graph, whose slope is
$$slope=\frac{\rho R}{M}$$
Since R and molar mass M are constant, slope is directly proportional to density:
$$slope∝ρ$$
From the graph, line corresponding to $$\rho_1$$ has greatest slope, then
$$\rho_2\ then\ \rho_3$$
Hence,
$$\rho_1>\rho_2>\rho_3$$

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
