Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
A thermodynamic system is taken through the cyclic process $$ABC$$ as shown in the figure. The total work done by the system during the cycle $$ABC$$ is ______ J.
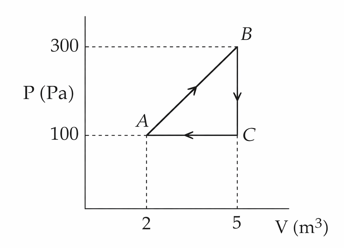
Correct Answer: 300
In a cyclic process on a P-V diagram, net work done equals area enclosed by the loop.
From graph:
The path ABCA forms a triangle.
Work done = area of triangle
$$W=\frac{1}{2}\times\text{base}\times\text{height}$$
Base (change in volume):
$$5-2=3\ \text{m}^3$$
Height (change in pressure):
$$300-100=200\ \text{Pa}$$
So,
$$W=\frac{1}{2}(3)(200)$$
$$W=300\ \text{J}$$

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
