Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
A thermodynamic system is taken from an original state $$A$$ to an intermediate state $$B$$ by a linear process as shown in the figure. Its volume is then reduced to the original value from $$B$$ to $$C$$ by an isobaric process. The total work done by the gas from $$A$$ to $$B$$ and $$B$$ to $$C$$ would be :
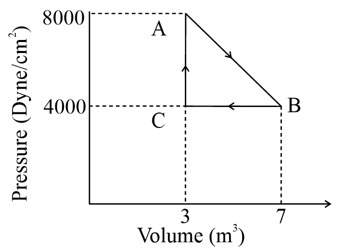
Work done is the area under the P-V curve.
From A→BA\to BA→B, pressure changes linearly, so work done is area of trapezium:
$$W_{AB}=\frac{P_A+P_B}{2}(V_B-V_A)$$
Given
$$P_A=8000\ \text{dyne/cm}^2,\quad P_B=4000\ \text{dyne/cm}^2$$
$$V_A=3m^3\ and\ V_B=7m^3$$
So
$$W_{AB}=\frac{8000+4000}{2}(7-3)$$
$$=6000\times4=24000$$
From B→C, process is isobaric compression, so
$$W_{BC}=P(V_C-V_B)$$
$$=4000(3−7)$$
=−16000
Therefore total work done,
$$W=W_{AB}+W_{BC}$$
$$=24000−16000=8000$$
Now converting units:
$$1\ \text{dyne/cm}^2=0.1\ \text{Pa}$$
So
$$8000\ \text{dyne/cm}^2=800\ \text{Pa}$$
Thus,
$$W=800(m^3)=800J$$
Create a FREE account and get:
Educational materials for JEE preparation
