Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
A sample of gas at temperature $$T$$ is adiabatically expanded to double its volume. Adiabatic constant for the gas is $$\gamma = 3/2$$. The work done by the gas in the process is: ($$\mu = 1$$ mole)
Find the work done when a gas at temperature $$T$$ is adiabatically expanded to double its volume with $$\gamma = 3/2$$ and $$\mu = 1$$ mole.
For an adiabatic process with an ideal gas, the relation $$TV^{\gamma-1} = \text{constant}$$ implies
$$ T_i V_i^{\gamma-1} = T_f V_f^{\gamma-1} $$
With $$V_f = 2V_i$$ and $$\gamma - 1 = 3/2 - 1 = 1/2$$, we have
$$ T \cdot V_i^{1/2} = T_f \cdot (2V_i)^{1/2} $$
$$ T_f = T \cdot \frac{V_i^{1/2}}{(2V_i)^{1/2}} = T \cdot \frac{1}{\sqrt{2}} = \frac{T}{\sqrt{2}} $$
For an adiabatic process, $$Q = 0$$, so the work done by the gas is $$W = -\Delta U$$. Equivalently, integrating $$W = \int P\,dV$$ with $$PV^\gamma = \text{const}$$ yields
$$W = \int P\,dV = \frac{P_iV_i - P_fV_f}{\gamma-1} = \frac{nRT_i - nRT_f}{\gamma-1} = \frac{nR(T_i-T_f)}{\gamma-1}$$
Substituting $$n = 1$$, $$T_i = T$$, $$T_f = T/\sqrt{2}$$, and $$\gamma - 1 = 1/2$$ gives
$$ W = \frac{1 \times R \times (T - T/\sqrt{2})}{1/2} = 2R \cdot T\left(1 - \frac{1}{\sqrt{2}}\right) = 2RT\left(\frac{\sqrt{2}-1}{\sqrt{2}}\right) = RT(2 - \sqrt{2}) $$
The correct answer is Option C: $$RT[2 - \sqrt{2}]$$.
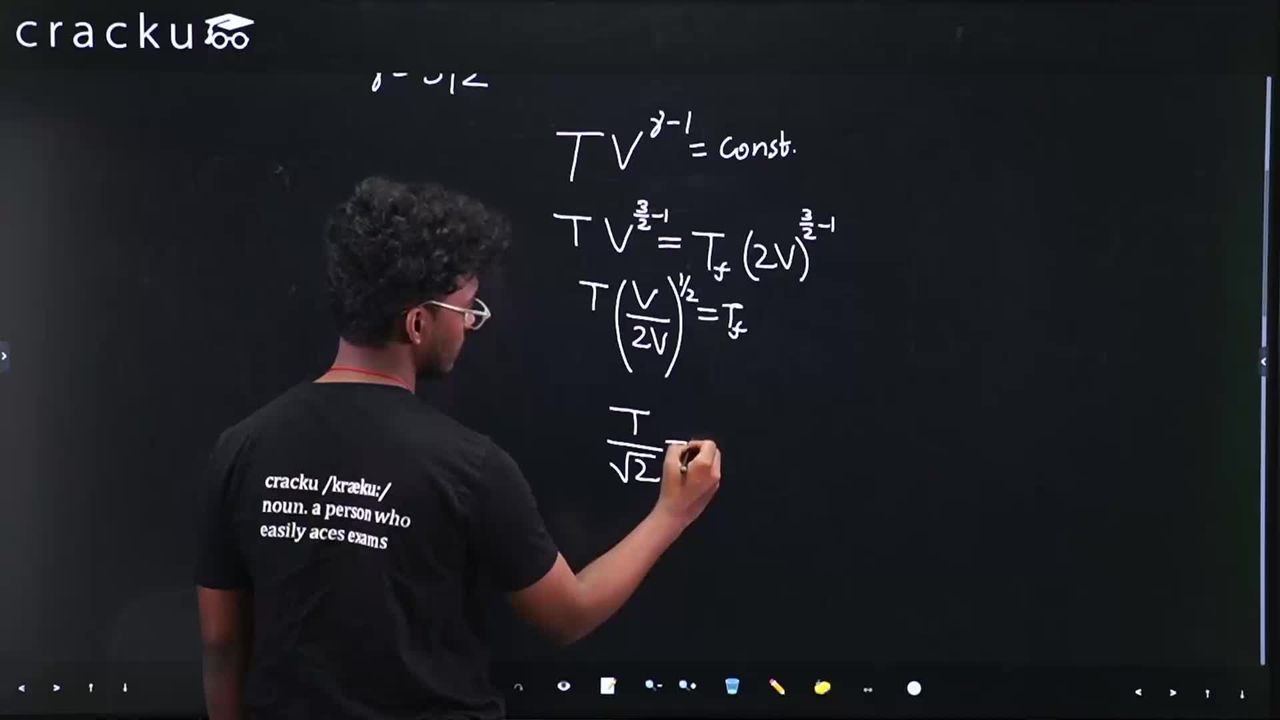
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
