Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
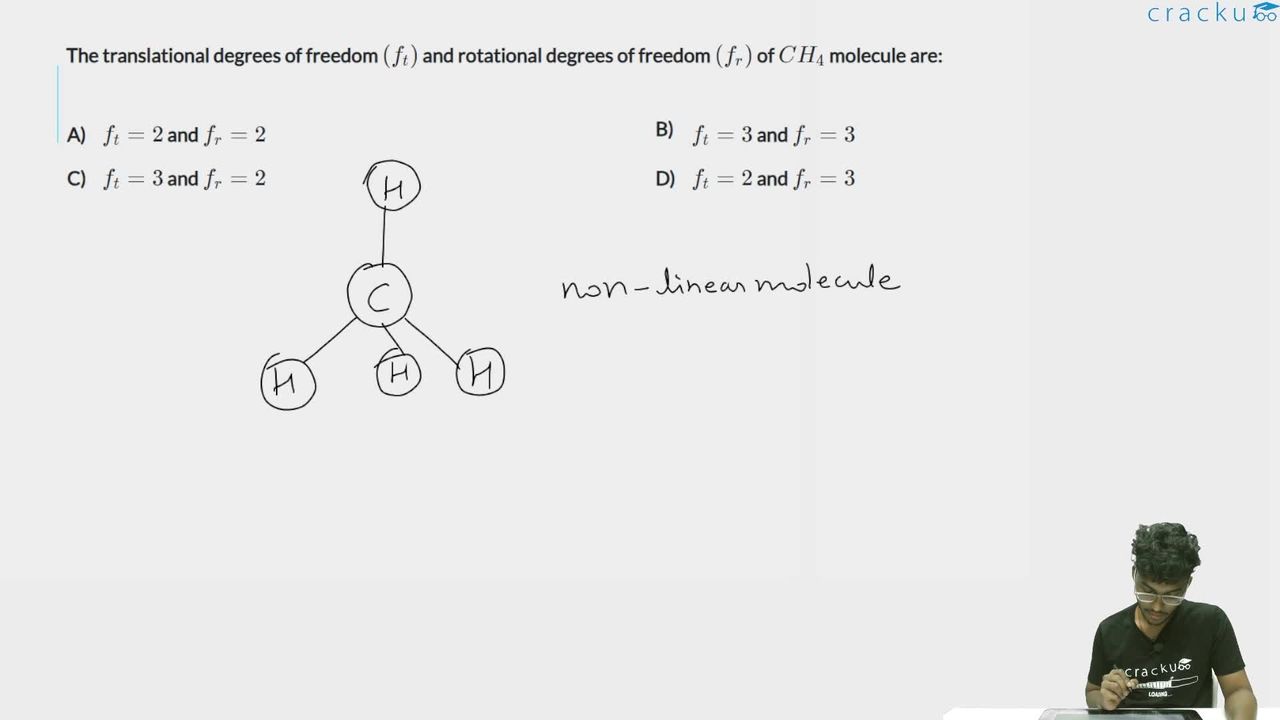
The translational degrees of freedom $$(f_t)$$ and rotational degrees of freedom $$(f_r)$$ of $$CH_4$$ molecule are:
Methane CH4 is a non-linear polyatomic molecule (tetrahedral structure).
For any molecule:
Translational degrees of freedom are always
$$f_t=3$$
because it can move along x,y,z directions.
For rotational degrees of freedom:
$$f_r=2$$
$$f_r=3$$
Since $$\text{CH}_4$$ is non-linear,
$$f_r=3$$
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
