Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Two thermodynamical processes are shown in the figure. The molar heat capacity for process $$A$$ and $$B$$ are $$C_A$$ and $$C_B$$. The molar heat capacity at constant pressure and constant volume are represented by $$C_P$$ and $$C_V$$, respectively. Choose the correct statement.
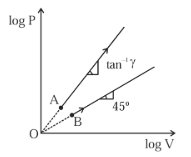
In the graph, the lines have positive slope in the (logV,logP) plane, so they represent
$$\log P=n\log V$$
or
$$P=V^n$$
which can be written as
$$PV^{-n}=\text{constant}$$This is a polytropic process with index
m=−n
For a polytropic process, molar heat capacity is
$$C=C_V+\frac{R}{1-m}$$Substituting m=−n,
$$C=C_V+\frac{R}{1+n}$$
For process A, slope is given by
$$\tanθ=γ$$
so
$$n=γ$$
Hence
$$C_A=C_V+\frac{R}{1+\gamma}$$
For process BBB, angle is $$45^{\circ}$$, so
n=1
and
$$C_B=C_V+\frac{R}{2}$$
Also,
$$C_P=C_V+R$$
Now comparing:
$$C_P>C_B$$
because
$$R>\frac{R}{2}$$
Also since γ>1,
$$\frac{R}{2}>\frac{R}{1+\gamma}$$
so
$$C_B>C_A$$
and clearly
$$C_A>C_V$$
Therefore,
$$CP>CB>CA>CV$$
Create a FREE account and get:
Educational materials for JEE preparation
