JEE Dual Nature of Matter & Radiation PYQs
JEE Dual Nature of Matter and Radiation PYQs are an important part of the JEE Physics syllabus. They help you understand what kind of questions are asked from this chapter and how well you know the main concepts, such as the dual nature of light, photoelectric effect, de Broglie wavelength, matter waves, work function, threshold frequency, and Einstein’s photoelectric equation.
In the exam, questions from this chapter usually come as direct numerical problems or simple concept-based questions. The good thing is that this chapter becomes much easier when your basics are clear. Once you understand the concepts properly and know which formula or idea to use, solving questions feels much more manageable. You do not need to think of this chapter as very difficult. With regular revision and smart practice, it can become one of the more scoring parts of JEE Physics.
In this blog, you will find a simple formula PDF, a section for important JEE Dual Nature of Matter and Radiation PYQs in download format, a few practice questions with answers, and some extra questions to solve on your own. You will also learn about common mistakes students often make and a few easy tips to save time in the exam.
JEE Dual Nature of Matter and Radiation Important PYQs PDF
This PDF can include the most important previous year questions from dual nature of matter and radiation. It may cover topics like wave and particle nature of light, photoelectric effect, work function, stopping potential, threshold frequency, Einstein’s photoelectric equation, de Broglie wavelength, and matter waves.
Practicing these questions will help you understand the exam pattern better. It will also improve your speed, accuracy, and confidence before the exam.
Important Formulas for JEE Dual Nature of Matter and Radiation PYQs
You only need a few important formulas and ideas to solve most dual nature of matter and radiation questions in JEE. These formulas help you understand photoelectric emission, energy relations, wavelength calculations, and matter-wave concepts more clearly.
You can download the full formula PDF from the link above. Here is a quick look at some of the main formulas:
Concept | Formula |
Energy of Photon | E = hν |
Relation Between Speed, Frequency, and Wavelength | c = νλ |
Einstein’s Photoelectric Equation | hν = ϕ + K.E. |
Maximum Kinetic Energy | K.E.max = hν − ϕ |
Work Function | ϕ = hν₀ |
Stopping Potential | eV₀ = K.E.max |
Threshold Frequency | ν₀ = ϕ / h |
de Broglie Wavelength | λ = h / p |
de Broglie Wavelength in Terms of Momentum | λ = h / mv |
de Broglie Wavelength for Electron | λ = h / √(2meV) |
Momentum of Photon | p = h / λ |
These formulas are commonly used in questions based on photons, photoelectric effect, stopping potential, de Broglie wavelength, and matter waves. If you revise them properly, many JEE questions start to feel much easier.
Top 5 Common Mistakes to Avoid in JEE Dual Nature of Matter and Radiation PYQs
Many students find this chapter confusing at first because it includes both light and matter concepts. But most mistakes happen because small details are missed while solving. Here are some common mistakes you should avoid:
Mixing up threshold frequency and stopping potential
Threshold frequency is the minimum frequency needed to remove an electron, while stopping potential is related to the maximum kinetic energy of the emitted electron. Many students confuse these two ideas.
Forgetting the role of work function
In photoelectric effect questions, the work function is very important. If you do not subtract it correctly from photon energy, the answer can go wrong.
Using the wrong formula for de Broglie wavelength
Students often remember the main formula but use the wrong form depending on whether momentum, mass, velocity, or potential is given.
Confusing intensity and frequency in photoelectric effect
The kinetic energy of emitted electrons depends on frequency, not on intensity. Intensity affects the number of emitted electrons, not their maximum kinetic energy.
Ignoring units
Always check units carefully. Values may be given in electron volt, joule, hertz, metre, or nanometre. A small unit mistake can change the whole answer.
List of JEE Dual Nature of Matter and Radiation PYQs
Here is a short set of JEE-style dual nature of matter and radiation questions for practice. These include common question types from photoelectric effect, photon energy, stopping potential, and de Broglie wavelength. Solving them regularly can help you become faster and more confident.
Question 1
A light wave described by $$E=60[\sin(3\times10^{15})t+\sin(12\times10^{15})t]$$ (in SI units) falls on a metal surface of work function 2.8 eV. The maximum kinetic energy of ejected photoelectron is (approximately) ___ eV. $$(h=6.6\times10^{-34}J.s\text{ and }e=1.6\times10^{19}C)$$
correct answer:- 4
Question 2
If an alpha particle with energy 7.7 MeV is bombarded on a thin gold foil, the closest distance from nucleus it can reach is ___ m. (Atomic number of gold = 79 and $$\frac{1}{4\pi\epsilon _{0}}=9\times10^{9} \text{in SI units} )$$
correct answer:- 1
Question 3
Light is incident on a metallic plate having work function $$110 \times 10^{-20}J$$. If the produced photoelectrons have zero kinetic energy then the angular frequency of the incident light is ___ rad/s. (h = $$6.63 \times 10^{-34}J.s.$$).
correct answer:- 1
Question 4
When a light of a given wavelength falls on a metallic surface the stopping potential for photoelectrons is 3.2 V. If a second light having wavelength twice of first light is used, the stopping potential drops to 0. 7 V. The wavelength of first light is ___ m.
$$(h= 6.63\times10^{-34}J.s,e=1.6\times10^{-19}C,c=3\times10^{8}m/s)$$
correct answer:- 2
Question 5
Number of photons of equal energy emitted per second by a 6 mW laser source operating at 663 nm is ____ . (Given: $$h=6.63\times 10^{-34}J.s\text{ and }c=3\times 10^{8} m/s$$)
correct answer:- 4
Question 6
light source of wavelength $$\lambda$$ illuminates a metal surface and electrons are ejected with maximum kinetic energy of 2 eV . If the same surface is illuminated by a light source of wavelength $$\frac{\lambda}{2}$$, then the maximum kinetic energy of ejected electrons will be (The work function of metal is 1 eV )
correct answer:- 4
Question 7
The work functions of cesium (Cs) and lithium (Li) metals are 1.9 eV and 2.5 eV , respectively. If we incident a light of wavelength 550 nm on these two metal surfaces, then photo-electric effect is possible for the case of
correct answer:- 3
Question 8
Two spherical bodies of same materials having radii 0.2 m and 0.8 m are placed in same atmosphere. The temperature of the smaller body is 800 K and temperature of the bigger body is 400 K . If the energy radiated from the smaller body is E, the energy radiated from the bigger body is (assume, effect of the surrounding temperature to be negligible),
correct answer:- 2
Question 9
In photoelectric effect an em-wave is incident on a metal surface and electrons are ejected from the surface. If the work function of the metal is 2.14 eV and stopping potential is 2 V , what is the wavelength of the emwave ? (Given hc = 1242eVnm where h is the Planck's constant and c is the speed of light in vaccum.)
correct answer:- 1
Question 10
In photoelectric effect, the stopping potential $$ (V_0)$$ $$v/s$$ frequency $$(\nu)$$ curve is plotted. ( $$h$$ is Planck's constant and $$\phi_0$$ is work function of the metal) $$(A) V_0$$ $$v/s \nu$$ is linear. $$(B)$$ } The slope of } $$V_0$$ $$v/s \nu$$ $$\text{ curve } = \frac{\phi_0}{h}\text{ (C) } h \text{ constant is related to the slope of the}$$ $$V_0$$ v/s $$\nu $$ line.(D) The value of electric charge of electron is not required to determine $$h$$ using the $$V_0$$ v/s $$\nu$$ curve. $$(E)$$ The work function can be estimated without knowing the value of h.Choose the correct answer from the options given below:
correct answer:- 2
Question 11
The ratio of the power of a light source $$S_1$$ to that of the light source $$S_2$$ is 2.$$S_1$$ is emitting $$2\times10^{15}$$ photons per second at 600,nm. If the wavelength of the source $$S_2$$ is 300, nm,then the number of photons per second emitted by $$S_2$$ is $$\underline {\hspace{2cm}}$$ $$\times 10^{14}.$$
correct answer:- 5
Question 12
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : Emission of electrons in photoelectric effect can be suppressed by applying a sufficiently negative electron potential to the photoemissive substance. Reason (R) : A negative electric potential, which stops the emission of electrons from the surface of a photoemissive substance, varies linearly with frequency of incident radiation. In the light of the above statements, choose the most appropriate answer from the options given below :
correct answer:- 4
Question 13
A sub-atomic particle of mass $$10^{-30}$$kg is moving with a velocity $$2.21\times10^{6}$$ m/s . Under the matter wave consideration, the particle will behave closely like
$$\left(h=6.63\times10^{-34}J.s\right)$$
correct answer:- 4
Question 14
The de Broglie wavelengths of a proton and an $$\alpha$$ particle are $$\lambda$$ and $$2\lambda$$ respectively. The ratio of the velocities of proton and $$\alpha$$ particle will be :
correct answer:- 4
Question 15
Monochromatic light of frequency $$6 \times 10^{14}$$ Hz is produced by a laser. The power emitted is $$2 \times 10^{-3}$$ W. How many photons per second on an average, are emitted by the source? (Given $$h = 6.63 \times 10^{-34}$$ J s)
correct answer:- 3
Question 16
A convex lens of focal length $$40$$ cm forms an image of an extended source of light on a photoelectric cell. A current $$I$$ is produced. The lens is replaced by another convex lens having the same diameter but focal length $$20$$ cm. The photoelectric current now is
correct answer:- 4
Question 17
The threshold frequency of a metal with work function 6.63 eV is :
correct answer:- 4
Question 18
The de-Broglie wavelength of an electron is the same as that of a photon. If velocity of electron is $$25\%$$ of the velocity of light, then the ratio of K.E. of electron and K.E. of photon will be:
correct answer:- 2
Question 19
Two sources of light emit with a power of $$200$$ W. The ratio of number of photons of visible light emitted by each source having wavelengths $$300$$ nm and $$500$$ nm respectively, will be:
correct answer:- 4
Question 20
The work function of a substance is $$3.0 \text{ eV}$$. The longest wavelength of light that can cause the emission of photoelectrons from this substance is approximately:
correct answer:- 2
Question 21
For the photoelectric effect, the maximum kinetic energy $$E_k$$ of the photoelectrons is plotted against the frequency $$(\nu)$$ of the incident photons as shown in figure. The slope of the graph give
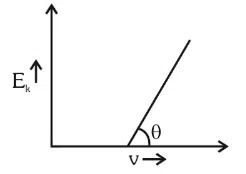
correct answer:- 4
Question 22
When a metal surface is illuminated by light of wavelength $$\lambda$$, the stopping potential is $$8$$ V. When the same surface is illuminated by light of wavelength $$3\lambda$$, stopping potential is $$2$$ V. The threshold wavelength for this surface is :
correct answer:- 3
Question 23
In a photoelectric effect experiment a light of frequency 1.5 times the threshold frequency is made to fall on the surface of photosensitive material. Now if the frequency is halved and intensity is doubled, the number of photo electrons emitted will be:
correct answer:- 3
Question 24
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R. Assertion A: Number of photons increases with increase in frequency of light. Reason R: Maximum kinetic energy of emitted electrons increases with the frequency of incident radiation. In the light of the above statements, choose the most appropriate answer from the options given below:
correct answer:- 3
Question 25
Which of the following statement is not true about stopping potential $$(V_0)$$ ?
correct answer:- 2
Question 26
In photoelectric experiment energy of $$2.48 \text{ eV}$$ irradiates a photo sensitive material. The stopping potential was measured to be $$0.5 \text{ V}$$. Work function of the photo sensitive material is :
correct answer:- 3
Question 27
Which of the following phenomena does not explain by wave nature of light. A. reflection B. diffraction C. photoelectric effect D. interference E. polarization. Choose the most appropriate answer from the options given below:
correct answer:- 3
Question 28
A proton and an electron have the same de Broglie wavelength. If $$K_p$$ and $$K_e$$ be the kinetic energies of proton and electron respectively, then choose the correct relation :
correct answer:- 2
Question 29
UV light of $$4.13 \text{ eV}$$ is incident on a photosensitive metal surface having work function $$3.13 \text{ eV}$$. The maximum kinetic energy of ejected photoelectrons will be:
correct answer:- 3
Question 30
A proton moving with one tenth of velocity of light has a certain de Broglie wavelength of $$\lambda$$. An alpha particle having certain kinetic energy has the same de-Broglie wavelength $$\lambda$$. The ratio of kinetic energy of proton and that of alpha particle is :
correct answer:- 2

 Group
Group






.webp)



















