Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
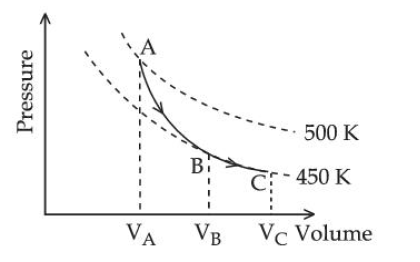
A poly-atomic molecule ($$C_V = 3R,\, C_P = 4R$$, where R is gas constant) goes from phase space point $$A\ (P_A = 10^5\,\text{Pa},\ V_A = 4 \times 10^{-6}\,\text{m}$$ to point $$B\ (P_B = 5 \times 10^4\,\text{Pa},\ V_B = 6 \times 10^{-6}\,\text{m}^3)$$ to point $$C\,(P_C = 10^4\,\text{Pa},\; V_C = 8 \times 10^{-6}\,\text{m}^3).$$ A to B is an adiabatic path B and C to is an isothermal path. The net heat absorbed per unit mole by the system is :
From the graph,$$A→B$$ is adiabatic, so no heat is exchanged in this part:
$$Q_{AB}=0$$
Hence net heat absorbed by the gas comes only during $$B→C$$, which is an isothermal expansion.
For one mole of an ideal gas in an isothermal process,
$$Q=W=RT\ln\frac{V_C}{V_B}$$
From the diagram, the path $$B→C$$ lies on the $$450K$$ isotherm, so
$$T=450K$$
Also,
$$\frac{V_C}{V_B}=\frac{8\times10^{-6}}{6\times10^{-6}}=\frac{4}{3}$$
Therefore,
$$Q=450R\ln\frac{4}{3}$$
Create a FREE account and get:
Educational materials for JEE preparation
