Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Two different adiabatic paths for the same gas intersect two isothermal curves as shown in P-V diagram. The relation between the ratio $$\frac{V_a}{V_d}$$ and the ratio $$\frac{V_b}{V_c}$$ is:
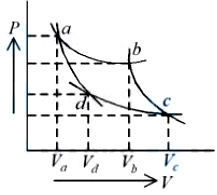
For an adiabatic process,
$$PV^{\gamma}=\text{constant}$$
There are two adiabatic curves:
For adiabatic through a and c:
$$P_aV_a^{\gamma}=P_cV_c^{\gamma}$$
So,
$$\frac{P_a}{P_c}=\frac{V_c^{\gamma}}{V_a^{\gamma}}$$
For adiabatic through b and d:
$$P_bV_b^{\gamma}=P_dV_d^{\gamma}$$
So,
$$\frac{P_b}{P_d}=\frac{V_d^{\gamma}}{V_b^{\gamma}}$$
Now a and b lie on same isotherm, so
$$P_aV_a=P_bV_b$$
$$\frac{P_a}{P_b}=\frac{V_b}{V_a}$$
Also c and d lie on same isotherm, so
$$P_cV_c=P_dV_d$$
$$\frac{P_c}{P_d}=\frac{V_d}{V_c}$$
Now divide the two adiabatic equations:
$$\frac{P_a/P_c}{P_b/P_d}=\left(\frac{\frac{V_C}{V_a}}{\frac{V_d}{V_b}}\right)^{\gamma}$$
Using isothermal relations,
$$\frac{(V_c/V_a)}{(V_d/V_b)}=\left(\frac{V_bV_c}{V_aV_d}\right)^{\gamma}$$
This simplifies only if
$$V_bV_c=V_aV_d$$
Hence,
$$\frac{V_a}{V_d}=\frac{V_b}{V_c}$$
Create a FREE account and get:
Educational materials for JEE preparation
