Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
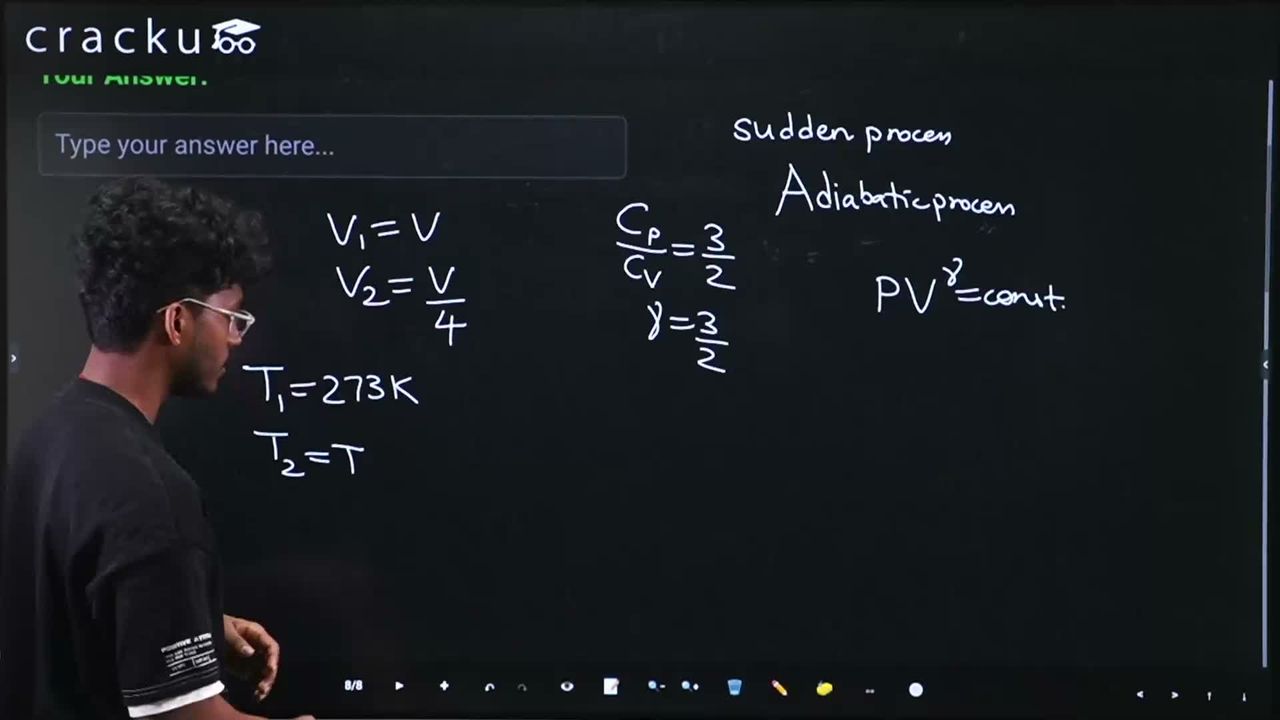
An ideal gas initially at $$0^{\circ}$$C temperature, is compressed suddenly to one fourth of its volume. If the ratio of specific heat at constant pressure to that at constant volume is 3/2, the change in temperature due to the thermodynamic process is _____ K.
Correct Answer: 273
For an adiabatic process (sudden compression), the relation between temperature and volume is $$ TV^{\gamma - 1} = \text{constant} $$. In this case, $$\gamma = \frac{C_p}{C_v} = \frac{3}{2}$$, the initial temperature is $$T_1 = 0°C = 273$$ K, and the gas is compressed to one-fourth of its original volume, so $$V_2 = \frac{V_1}{4}$$.
Applying $$ T_1 V_1^{\gamma - 1} = T_2 V_2^{\gamma - 1} $$ gives $$ T_2 = T_1 \left(\frac{V_1}{V_2}\right)^{\gamma - 1} = 273 \times (4)^{3/2 - 1} = 273 \times 4^{1/2} = 273 \times 2 = 546 \text{ K} $$.
Therefore, the change in temperature is $$ \Delta T = T_2 - T_1 = 546 - 273 = 273 \text{ K} $$. The temperature rise is 273 K.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
