Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
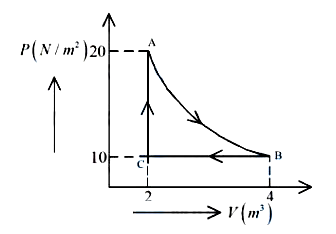
A real gas within a closed chamber at $$27°C$$ undergoes the cyclic process as shown in figure. The gas obeys $$PV^3 = RT$$ equation for the path $$A$$ to $$B$$. The net work done in the complete cycle is (assuming $$R = 8 \text{ J/molK}$$):
or a cyclic process, net work done is the sum of work in each path.
Cycle is
$$C→A→B→C$$
For C→A, volume is constant, so
$$W_{CA}=0$$
For A→B, given
$$PV^3=RT$$
Temperature is
$$27^{\circ}C=300K$$
and
R=8
So
$$PV^3=8(300)=2400$$
Thus
$$P=\frac{2400}{V^3}$$
Work done from A to B is
$$W_{AB}=\int PdV$$
$$=\int_2^4\frac{2400}{V^3}dV$$
$$=2400\int_2^4V^{-3}dV$$
$$=2400\left(\frac{-1}{2V^2}\right)_2^4$$
$$2400\left(\frac{1}{8}-\frac{1}{32}\right)$$
$$=2400\cdot\frac{3}{32}$$
=225J
For B→C, pressure is constant at
P=10
So
$$W_{BC}=P(V_C-V_B)$$=10(2−4)
=−20J
Hence net work done in the complete cycle is
W=0+225−20W
=205J
Create a FREE account and get:
Educational materials for JEE preparation
