Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
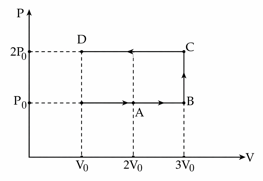
Using the given P - V diagram, the work done by an ideal gas along the path ABCD is :
Path is:
$$A(2V_0,P_0)→B(3V_0,P_0)→C(3V_0,2P_0)→D(V_0,2P_0)$$
Work done is
$$W=\int PdV$$
Calculate segment-wise.
Along AB (constant pressure $$P_0$$):
$$W_{AB}=P_0(3V_0-2V_0)=P_0V_0$$
Along BC (constant volume):
$$W_{BC}=0$$
Along CD (constant pressure $$2P_0$$ compression):
$$W_{CD}=2P_0(V_0-3V_0)$$
$$=2P_0(-2V_0)=-4P_0V_0$$
Total work:
$$W=W_{AB}+W_{BC}+W_{CD}$$
$$P_0V_0-4P_0V_0$$
$$=-3P_0V_0$$
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
