JEE Chemical Bonding PYQs
JEE Chemical Bonding PYQs are an important part of the JEE Chemistry syllabus. They help you understand the kind of questions asked from this chapter and test how well you know the basic concepts, such as ionic bond, covalent bond, coordinate bond, VSEPR theory, hybridization, bond angle, molecular shape, and polarity.
In the exam, chemical bonding questions can appear as direct theory-based questions or as concept-based problems. The good thing is that this chapter becomes much easier when your basics are clear. Once you understand the concepts properly and know which rule or idea to use, solving questions feels much simpler. You do not need to think of chemical bonding as a difficult chapter. With regular practice and proper revision, it can become one of the more scoring topics in Chemistry.
In this blog, you will find a simple formula PDF, a section for important JEE Chemical Bonding PYQs in download format, a few practice questions with answers, and some extra questions to solve on your own. You will also learn about common mistakes students often make and a few easy tips to save time in the exam.
JEE Chemical Bonding Important PYQs PDF
This PDF can include the most important previous year questions from chemical bonding. It may cover topics like ionic bond, covalent bond, Lewis structures, formal charge, VSEPR theory, hybridization, molecular geometry, bond order, resonance, dipole moment, and hydrogen bonding.
Practicing these questions will help you understand the exam pattern better. It will also improve your speed, accuracy, and confidence before the exam.
Important Formulas for JEE Chemical Bonding PYQs
You only need a few important formulas and rules to solve most chemical bonding questions in JEE. These help you find bond order, formal charge, dipole moment, and understand molecular shape and stability more easily.
You can download the full formula PDF from the link above. Here is a quick look at some of the main formulas and rules:
Concept | Formula / Rule |
Formal Charge | Formal Charge = Valence Electrons − Lone Pair Electrons − ½(Bonding Electrons) |
Bond Order (MOT) | Bond Order = (Number of Bonding Electrons − Number of Antibonding Electrons) / 2 |
Dipole Moment | μ = q × d |
Octet Rule | Atoms tend to complete 8 electrons in valence shell |
Duplet Rule | Hydrogen becomes stable with 2 electrons |
Sigma Bond | Formed by head-on overlap |
Pi Bond | Formed by sidewise overlap |
sp Hybridization | 2 hybrid orbitals, linear shape |
sp² Hybridization | 3 hybrid orbitals, trigonal planar shape |
sp³ Hybridization | 4 hybrid orbitals, tetrahedral shape |
These formulas and rules are commonly used in questions based on molecular structure, bond strength, resonance, hybridization, polarity, and bond order. If you revise them properly, many JEE questions will feel much easier to solve.
Top 5 Common Mistakes to Avoid in JEE Chemical Bonding PYQs
Many students find chemical bonding confusing, but in most cases, the real problem is not the chapter itself. It is the small mistakes made while solving. Here are some common mistakes you should avoid:
Drawing the wrong Lewis structure
A small mistake in placing electrons can change the whole structure. Always count valence electrons carefully before drawing the final structure.
Confusing molecular shape with electron pair geometry
Many students mix up the actual shape of the molecule with the arrangement of electron pairs. Lone pairs can change the final molecular shape.
Using the wrong hybridization
Hybridization depends on the number of bond pairs and lone pairs around the central atom. If you count them incorrectly, the answer can go wrong.
Ignoring formal charge and resonance
Sometimes more than one structure is possible. In such cases, formal charge and resonance help you choose the most stable one.
Not checking polarity properly
Some students look only at bond polarity and forget to check the overall shape of the molecule. Because of this, they give the wrong answer for molecular polarity.
List of JEE Chemical Bonding PYQs
Here is a short set of JEE-style chemical bonding questions for practice. These include common question types from Lewis structure, formal charge, hybridization, bond order, and polarity. Solving them regularly can help you become faster and more confident.
Question 1
Given below are two statements:
Statement I: The correct order in terms of bond dissociation enthalpy is $$Cl_{2} > Br_{2} > F_{2} > I_{2}$$.
Statement II : The correct trend in the covalent character of the metal halides is $$[SnCl_{4} > SnCl_{2}]$$, $$[PbCl_{4}> PbCl_{2}]$$, and $$[UF_{4} > UF_{6}]$$.
In The light oh the above statements, choose the correct answer from the options given below:
correct answer:- 3
Question 2
The correct increasing order of C - H(A), C - 0 (B), C = O(C) and C = N (D) bonds in terms of covalent bond length is :
correct answer:- 1
Question 3
Among $$H_{2}S$$, $$H_{2}O$$, $$NF_{3}$$, $$NH_{3}$$ and $$CHC1_{3}$$, identify the molecule (X) with lowest dipole moment value. The number of lone pairs of electrons present on the central atom of the molecule (X) is :
correct answer:- 4
Question 4
ldentify the molecule (X) with maximum number of lone pairs of electrons (obtained using Lewis dot structure) among $$HNO_{3},H{2}SO_{4},NF_{3}\text{ and }O_{3}$$. Choose the correct bond angle made by the central atom of the molecule (X).
correct answer:- 2
Question 5
The formal charges on the atoms marked as (1) to (4) in the Lew is representation
of $$HNO_{3}$$ molecule respectively are
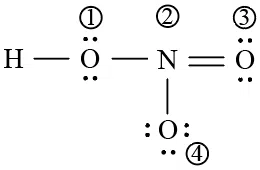
correct answer:- 1
Question 6
Consider the transition metal ions $$Mn^{3+}, Cr^{3+}, Fe ^{3+}$$ and $$Co^{3+}$$ and all form low spin octahedral complexes. The correct decreasing order of unpaired electrons in their respective d-orbitals of the complexes is
correct answer:- 3
Question 7
Given below are statements about some molecules/ions.
Identify the CORRECT statements.
A The dipole moment value of $$NF_{3}$$ is higher than that of $$NH_{3}$$.
B. The dipole moment value of $$BeH_{2}$$ is zero.
C. The bond order of $$O_{2}^{2-}$$ and $$F_{2}$$ is same.
D. The formal charge on the central oxygen atom of ozone is -1 .
E. In $$NO_{2}$$, all the three atoms satisfy the octet rule, hence it is very stable.
Choose the correct answer from the options given below:
correct answer:- 3
Question 8
Find out the statements which are not true.
A. Resonating structures with more number of covalent bonds and lesser charge separntion are more stable.
B. In electromeric effect, an unsaturated system shows +E effect with nucleophile and -E effect with electrophile.
C. Inductive effect is responsible for high melting point, boiling point and dipole moment of polar compotmds.
D. The greater the number of alkyl groups attached to the doubly bonded carbon atoms, higher is the heat of hydrogenation.
E. Stability of carbanion increases with the increase in s - character of the carbon carrying the negative charge.
Choose the correct answer from the options given below:
correct answer:- 4
Question 9
Pair of species among the following having same bond order as well as paramagnetic character will be-
correct answer:- 2
Question 10
Given below are two statements:
Statement I: The number of species among $$BF_{4}^{-},SiF_{4},XeF_{4}\text{ and }SF_{4}$$,that have unequal E-F bond lengths is two. Here, E is the central atom.
Satement II: Among $$O_{2}^{-},O_{2}^{2-},F_{2}\text{ and }O_{2}^{+},O_{2}^{-}$$ has the highest bond order.
In the light of the above statements, choose the correct answer from the options given below
correct answer:- 4
Question 11
Which statements are NOT TRUE about $$XeO_2 F_2$$?
A. It has a see-saw shape.
B. Xe has 5 electron pairs in its valence sheU in XeO 2 F 2.
C. The $$O - Xe- O$$ bond angle is close to $$180^{o}$$.
D. The $$F- Xe -F$$ bond angle is close to $$180^{o}$$.
E. $$Xe$$ has 16 valence electrons in $$XeO_2 F_2$$.
Choose the correct answer from the options given below:
correct answer:- 1
Question 12
Arrange the following compounds in increasing order of their dipole moment : $$HBr,H_{2}S,NF_{3}\text{ and }CHCl_{3}$$
correct answer:- 2
Question 13
The maximum covalency of a non-metallic group 15 element ' E ' with weakest E-E bond is:
correct answer:- 1
Question 14
The number of molecules/ions that show linear geometry among the following is ______
$$ \mathrm{SO_2},\ \mathrm{BeCl_2},\ \mathrm{CO_2},\ \mathrm{N_3^-},\ \mathrm{NO_2},\ \mathrm{F_2O},\ \mathrm{XeF_2},\ \mathrm{NO_2^+},\ \mathrm{I_3^-},\ \mathrm{O_3} $$
correct answer:- 6
Question 15
Which of the following statement is true with respect to $$H_2O,\ NH_3$$ and $$CH_4?$$ $$A.$$ The central atoms of all the molecules are $$sp^3$$ hybridized. $$B.$$ The $$H-O-H,\ H-N-H$$ and $$H-C-H$$ angles in the above molecules are $$104.5^\circ,\ 107.5^\circ$$ and $$109.5^\circ$$ respectively. $$C.$$ The increasing order of dipole moment is $$CH_4 < NH_3 < H_2O.$$ $$D.$$ Both $$H_2O$$ and $$NH_3$$ are Lewis acids and $$CH_4$$ is a Lewis base. $$E.$$ A solution of $$NH_3$$ in $$H_2O$$ is basic. In this solution $$NH_3$$ and $$H_2O$$ act as Lowry-Bronsted acid and base respectively. Choose the correct answer from the options given below:
correct answer:- 1
Question 16
Which of the following linear combination of atomic orbitals will lead to formation of molecular orbitals in homonuclear diatomic molecules [internuclear axis in $$z$$-direction] ? A. $$2p_z$$ and $$2p_x$$ B. 2 s and $$2p_x$$ C. 3 $$d_{xy}$$ and 3 $$d_{x^{2} - y^{2}}$$ D. 2 s and $$2p_z$$ E. $$2p_z$$ and $$3d_{x}^{2}- y^{2}$$ Choose the correct answer from the options given below:
correct answer:- 2
Question 17
Given below are two statements : Statement (I): Experimentally determined oxygen-oxygen bond lengths in the $$ O_3 $$ are found to be same and the bond length is greater than that of a $$O = O$$ (double bond) but less than that of a single $$(O - O)$$ bond. Statement (II) : The strong lone pair-lone pair repulsion between oxygen atoms is solely responsible for the fact that the bond length in ozone is smaller than that of a double bond $$(O = O)$$ but more than that of a single bond $$(O - O)$$. In the light of the above statements, choose the correct answer from the options given below :
correct answer:- 3
Question 18
The molecules having square pyramidal geometry are
correct answer:- 4
Question 19
Consider $$'n'$$ is the number of lone pair of electrons present in the equatorial position of the most stable structure of $$ClF_{3}$$. The ions from the following with $$'n'$$ number of unpaired electrons are $$A. V^{3+} B.Ti^{3+} C.Cu^{2+} D.Ni^{2+} E.Ti^{2+}$$ Choose the correct answer from the options given below :
correct answer:- 2
Question 20
Match the LIST-I with LIST-II
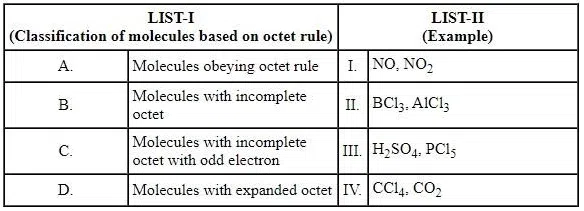
Choose the correct answer from the options given below:
correct answer:- 2
Question 21
Total number of sigma ($$\sigma$$) and pi($$\pi$$) bonds respectively present in hex-1-en-4-yne are :
correct answer:- 3
Question 22
Total number of non bonded electrons present in $$NO_{2}$$ -ion based on Lewis theory is
correct answer:- 12
Question 23
Which one of the following molecules has maximum dipole moment?
correct answer:- 4
Question 24
Number of molecules/ions from the following in which the central atom is involved in sp³ hybridization is: $$NO_3^-,\ BCl_3,\ ClO_2^-,\ ClO_3$$
correct answer:- 3
Question 25
Number of molecules/species from the following having one unpaired electron is ______. $$O_2,\ O_2^{-1},\ NO,\ CN^{-1},\ O_2^{2-}$$
correct answer:- 2
Question 26
Total number of molecules/species from following which will be paramagnetic is ______
$$O_{2},O_2^+,O_2^-,NO,NO_{2},CO,K_{2}[NiCl_{4}],[Co(NH_{3})_{6}]Cl_{3},K_{2}[Ni(CN)_{4}]$$
correct answer:- 6
Question 27
Arrange the bonds in order of increasing ionic character in the molecules: $$LiF$$, $$K_2O$$, $$N_2$$, $$SO_2$$ and $$ClF_3$$.
correct answer:- 3
Question 28
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R. Assertion A: $$PH_3$$ has lower boiling point than $$NH_3$$. Reason R: In liquid state $$NH_3$$ molecules are associated through Vander Waal's forces, but $$PH_3$$ molecules are associated through hydrogen bonding. In the light of the above statements, choose the most appropriate answer from the options given below:
correct answer:- 4
Question 29
The number of molecules/ion/s having trigonal bipyramidal shape is: $$PF_5$$, $$BrF_5$$, $$PCl_5$$, $$[PtCl_4]^{2-}$$, $$BF_3$$, $$Fe(CO)_5$$
correct answer:- 3
Question 30
Given below are two statements:
Statement (I): A $$\pi$$ bonding MO has lower electron density above and below the inter-nuclear axis.
Statement (II): The $$\pi^*$$ antibonding MO has a node between the nuclei.
In light of the above statements, choose the most appropriate answer from the options given below:
correct answer:- 3
Question 31
Select the compound from the following that will show intramolecular hydrogen bonding.
correct answer:- 4
Question 32
Choose the polar molecule from the following :
correct answer:- 4
Question 33
Sum of bond order of CO and $$NO^+$$ is _______.
correct answer:- 6
Question 34
The number of non-polar molecules from the following is
$$HF, H_2O, SO_2, H_2, CO_2, CH_4, NH_3, HCl, CHCl_3, BF_3$$
correct answer:- 4
Question 35
Number of compounds with one lone pair of electrons on central atom amongst following is _______
$$O_3, H_2O, SF_4, ClF_3, NH_3, BrF_5, XeF_4$$
correct answer:- 4
Question 36
The number of species from the following which are paramagnetic and with bond order equal to one is _______
$$H_2, He_2^+, O_2^+, N_2^{2-}, O_2^{2-}, F_2, Ne_2^+, B_2$$
correct answer:- 1
Question 37
The total number of molecules with zero dipole moment among $$CH_4, BF_3, H_2O, HF, NH_3, CO_2$$ and $$SO_2$$ is ______.
correct answer:- 3
Question 38
The total number of anti bonding molecular orbitals, formed from $$2s$$ and $$2p$$ atomic orbitals in a diatomic molecule is ______.
correct answer:- 4
Question 39
The total number of molecular orbitals formed from $$2s$$ and $$2p$$ atomic orbitals of a diatomic molecule is _______.
correct answer:- 8
Question 40
Given below are two statements:
Statement-I: Since fluorine is more electronegative than nitrogen, the net dipole moment of $$NF_3$$ is greater than $$NH_3$$.
Statement-II: In $$NH_3$$, the orbital dipole due to lone pair and the dipole moment of NH bonds are in opposite direction, but in $$NF_3$$ the orbital dipole due to lone pair and dipole moments of N-F bonds are in same direction.
In the light of the above statements, choose the most appropriate from the options given below.
correct answer:- 2
Question 41
The molecule/ion with square pyramidal shape is:
correct answer:- 3
Question 42
The linear combination of atomic orbitals to form molecular orbitals takes place only when the combining atomic orbitals A. have the same energy B. have the minimum overlap C. have same symmetry about the molecular axis D. have different symmetry about the molecular axis. Choose the most appropriate from the options given below:
correct answer:- 2
Question 43
The number of species from the following in which the central atom uses $$sp^3$$ hybrid orbitals in its bonding is _________. $$NH_3, SO_2, SiO_2, BeCl_2, CO_2, H_2O, CH_4, BF_3$$
correct answer:- 4
Question 44
Which of the following is least ionic?
correct answer:- 2
Question 45
A diatomic molecule has a dipole moment of 1.2 D. If the bond distance is 1 $$\mathring{A}$$, then fractional charge on each atom is ______ $$\times 10^{-1}$$ esu. (Given 1D = $$10^{-18}$$ esu cm)
correct answer:- 0
Question 46
The correct statement/s about Hydrogen bonding is/are: A. Hydrogen bonding exists when H is covalently bonded to the highly electronegative atom. B. Intermolecular H bonding is present in o-nitrophenol. C. Intramolecular H bonding is present in HF. D. The magnitude of H bonding depends on the physical state of the compound. E. H-bonding has powerful effect on the structure and properties of compounds. Choose the correct answer from the options given below:
correct answer:- 2
Question 47
The number of species from the following that have pyramidal geometry around the central atom is _______. $$S_2O_3^{2-}$$, $$SO_4^{2-}$$, $$SO_3^{2-}$$, $$S_2O_7^{2-}$$
correct answer:- 4
Question 48
Number of compounds/species from the following with non-zero dipole moment is ______. $$BeCl_2, BCl_3, NF_3, XeF_4, CCl_4, H_2O, H_2S, HBr, CO_2, H_2, HCl$$
correct answer:- 5
Question 49
Match List I with List II :

Choose the correct answer from the options given below :
correct answer:- 1
Question 50
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : $$NH_3$$ and $$NF_3$$ molecule have pyramidal shape with a lone pair of electrons on nitrogen atom. The resultant dipole moment of $$NH_3$$ is greater than that of $$NF_3$$. Reason (R) : In $$NH_3$$, the orbital dipole due to lone pair is in the same direction as the resultant dipole moment of the $$N-H$$ bonds. $$F$$ is the most electronegative element. In the light of the above statements, choose the correct answer from the options given below :
correct answer:- 1

 Group
Group






.webp)














