Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Let us analyze each molecule for polarity:
- $$CCl_4$$: Tetrahedral, symmetric, dipole moments cancel. Non-polar.
- $$CO_2$$: Linear, symmetric, dipole moments cancel. Non-polar.
- $$CH_2=CH_2$$: Planar, symmetric. Non-polar.
- $$CHCl_3$$: Tetrahedral but asymmetric (one H, three Cl). Net dipole moment exists. Polar.
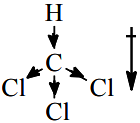
The answer is $$CHCl_3$$, which corresponds to Option (4).
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
