JEE Electrochemistry PYQs
The JEE Chemistry syllabus also includes JEE Electrochemistry PYQs. They assist you realize what type of questions were requested of this chapter and how familiar you are with the primary subjects, which are redox reactions, electrolytic conductance, electrochemical cells, electrode potential, Nernst equation and the laws created by Faraday.
Questions in the exam about electrochemistry are typically in the form of direct numerical questions or in the form of simple concept based questions. The only advantage is that this chapter is much easier when you understand your basics. After getting the concepts in the right way and getting to know which formula or rule to apply, then it is much easier to solve the questions. Electrochemistry is a chapter you do not have to find very difficult. It may become one of the higher scoring sections of JEE Chemistry with frequent revision and intelligent practice.
In this blog, you will find a simple formula PDF, a section for important JEE Electrochemistry PYQs in download format, a few practice questions with answers, and some extra questions to solve on your own. You will also learn about common mistakes students often make and a few simple tips to save time in the exam.
JEE Electrochemistry Important PYQs PDF
This PDF is able to contain the most significant past year questions of electrochemistry. It can include redox reactions, galvanic cells, electrolytic cells, standard electrode potential, Nernst equation, conductivity, molar conductivity, laws of electrolysis describing Faraday and the electrochemical series.
These questions will help you to be more familiar with the exam pattern. It will also enhance your speed, accuracy and confidence prior to the exams.
Important Formulas for JEE Electrochemistry PYQs
You only need a few important formulas and ideas to solve most electrochemistry questions in JEE. These formulas help you understand cell reactions, potential difference, conductivity, and electrolysis calculations more clearly.
You can download the full formula PDF from the link above. Here is a quick look at some of the main formulas:
Concept | Formula |
Cell Potential | Ecell = Ecathode − Eanode |
Nernst Equation | E = E° − (0.0591/n) log Q |
Gibbs Energy Relation | ΔG = −nFE |
Standard Gibbs Energy Relation | ΔG° = −nFE° |
Equilibrium Constant Relation | E° = (0.0591/n) log K |
Conductance | G = 1/R |
Specific Conductivity | κ = Cell Constant × Conductance |
Molar Conductivity | Λm = κ × 1000 / C |
Faraday’s First Law | Mass deposited ∝ quantity of electricity |
Faraday’s Second Law | Mass deposited ∝ equivalent mass |
Charge | Q = It |
These formulas are commonly used in questions based on electrochemical cells, conductivity, electrode potential, electrolysis, and redox reactions. If you revise them properly, many JEE questions start to feel much easier.
Top 5 Common Mistakes to Avoid in JEE Electrochemistry PYQs
Many students find electrochemistry confusing in the beginning because it has both concepts and calculations. But in most cases, mistakes happen because small details are missed while solving. Here are some common mistakes you should avoid:
Mixing up anode and cathode
Students often get confused about where oxidation and reduction take place. Always remember that oxidation happens at the anode and reduction happens at the cathode.
Using the wrong sign in cell potential
While calculating cell potential, the correct sign is very important. Even a small mistake in electrode potential can change the whole answer.
Forgetting the value of n in the Nernst equation
In many questions, students remember the formula but forget to use the correct number of electrons transferred in the reaction.
Confusing conductance and conductivity
Conductance depends on the conductor, while conductivity is a property of the material or solution. They are related, but they are not the same.
Making mistakes in electrolysis calculations
In Faraday’s law questions, students often forget to convert current, time, or charge properly. A simple unit mistake can give the wrong final answer.
List of JEE Electrochemistry PYQs
Here is a short set of JEE-style electrochemistry questions for practice. These include common question types from cell potential, conductivity, the Nernst equation, and electrolysis. Solving them regularly can help you become faster and more confident.
Question 1
For a closed circuit Daniell cell, which of the following plots is the accurate one at a given temperature?
correct answer:- 2
Question 2
MX is a sparingly soluble salt that follows the given solubility equilibrium at 298 K.
$$MX(s) \rightleftharpoons M^{+} (aq)+X^{-}(aq); K_{sp}=10^{-10}$$
If the standard reduction potential for $$M^{+}(aq)\xrightarrow {+e^{-}}M(s)$$ and $$\left(E_{M^{+}/M}^\ominus\right)=0.79V$$, then the value of the standard reduction potential for the metal/metal insoluble salt electrode $$E^{\ominus}_{X^{-}/MX (s)/M }$$ is __________mV.(nearest integer)
[Given : $$\frac{2.303RT}{F} = 0.059V$$]
correct answer:- 200
Question 3
The pH and conductance of a weak acid (HX) was found to be 5 and $$4\times10^{5}S$$. respectively. The conductance was measured under standard condition using a cell where the electrode plates having a surface area of 1 $$cm^{2}$$ were at a distance of 15 cm apart. The value of the limiting molar conductivity is ______ S $$m^{2}mol^{-1}$$ (nearest integer)
(Given : degree of dissociation of the weak acid ($$\alpha$$) < < 1)
correct answer:- 6
Question 4
Consider the following electrochemical cell : $$Pt| O_{2}(g)(1 bar) | HCl(aq)||M^{2+}(aq,1.0M)|M(s)$$
The pH above which , oxygen gas would start to evolve at anode is ____ (nearest integer).
[Given: $$E_{M^{2+}/m}^\circ = 0.994V$$ , $$E_{O^{2}/H_{2}O}^\circ = 1.23V$$ are standard reduction potential and $$\frac{RT}{F}(2.303)=0.059V$$ at the given condition]
correct answer:- 4
Question 5
In the given electrochemical cell, $$Ag(s)|AgCl(s)|FeCl_{2}(aq),FeCl_{3}(aq)|Pt(s)$$ at298 K, theceU potential $$E_{cell}$$ will increase when:
A. Concentration of $$Fe^{2+}$$ is increased.
B. Concentration of $$Fe^{3+}$$ is decreased.
C. Concentration of $$Fe^{2+}$$ is decreased.
D. Concentration of $$Fe^{3+}$$ is increased.
E. Concentration of $$Cl^{-}$$ is increased.
Choose the correct answer from the options given below :
correct answer:- 3
Question 6
Consider the following electrochemical cell at 298K
$$ Pt \mid HSnO_2{^-}(aq)\mid Sn(OH)_6{^{2-}}(aq)\mid OH^{-}(aq)\mid Bi_{2}O_{3}(s)\mid Bi(s)$$.
If the reaction quotient at a given time is $$10^{6}$$, then the cell $$EMF (E_{cell})$$ is _____ $$\times 10^{-1} V$$ (Nearest integer).
Given the standard half-cell reduction potential as
$$E_{Bi_{2}O_{3}/Hi,OH^{-}}^{o}=-0.44V \text{ and }E_{Sn(OH)_6^{2-}/HSnO_2^{-}, OH^{-}}^{o}=-0.90V$$
correct answer:- 4
Question 7
Electricity is passed through an acidic solution of $$Cu^{2+}$$ till all the Cu2+ was exhausted, leading to the deposition of 300 mg of Cu metal. However, a current of 600 mA was continued to pass through the same solution for another 28 minutes by keeping the total volume of the solution fixed at 200 mL. The total volume of oxygen evolved at STP during the entire process is __ mL. (Nearest integer)
[Given:
$$Cu^{2+}(aq)+2e^{-}\rightarrow Cu(s) E_{red}^{\circ}=+0.34V$$
$$O_{2}(g)+4H^{+}+4e^{-}\rightarrow 2H_{2}O E_{red}^{\circ}=+1.23V$$
Molar mass of Cu= 63.54 g $$mol^{-1}$$
Molar mass of $$O_{2}$$ = 32 g $$mol^{-1}$$
Farnday Constant = 96500 C $$mol^{-1}$$
Molar volume at STP = 22.4 L]
correct answer:- 111
Question 8
Consider the following redox reaction taking place in acidic medium
$$BH_{4}^{-}(aq)+ClO_{3}^{-}(aq)\rightarrow H_{2}BO_{3}^{-}(aq)+Cl^{-}(aq)$$
If the Nerst equation for the above balanced reaction is $$E_{cell}=E_{cell}^{\circ}-\frac{RT}{nF}ln Q$$, then the value of n is______.(Nearest integer)
correct answer:- 24
Question 9
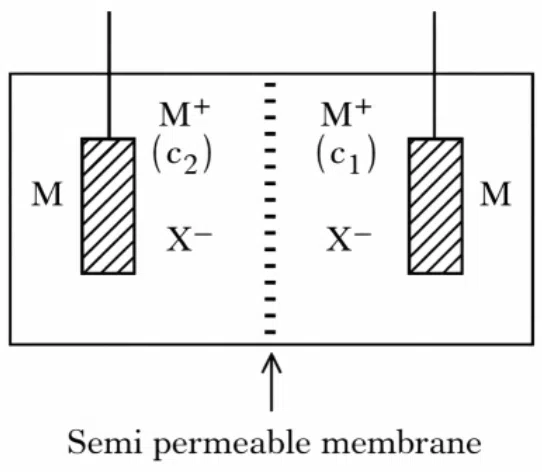
Consider the above electrochemical cell where a metal electrode (M) is undergoing redox reaction by forming $$M^{+}(M\rightarrow M^{+}+e^{-})$$. The cation $$M^{+}$$ is present in two different concentrations $$c_{1}$$ and $$c_{2}$$ as shown above. which of the following statement is correct for generating a positive cell potential?
correct answer:- 2
Question 10
The plot of $$\log_{10}^{K} VS \frac{1}{T}$$ gives a straight Line. The intercept and slope respectively are
(where K is equilibrium constant).
correct answer:- 2
Question 11
A volume of x mL of 5 M $$NaHCO_{3}$$ solution was mixed with 10 mL of 2 M $$H_{2}CO_{3}$$ solution to make an electrolytic buffer. If the same buffer was used in the following electrochemical cell to record a cell potential of 235.3 mV, then the value of x=_______ mL (nearest integer).
$$Sn(s)|Sn(OH)_{6}^{2-}(0.5 M)|HSnO_{2}^{-}(0.05 M)|OH^{-}|Bi_{2}O_{3}(s)|Bi(s)$$
Consider upto one place of decimal for intermediate calculations

correct answer:- 78
Question 12
For strong electrolyte $$\lambda_m$$ increases slowly with dilution and can be represented by the equation
$$\Lambda_m = \Lambda_m^\circ - A c^{1/2}$$
Molar conductivity values of the solutions of strong electrolyte AB at 18°C are given below:
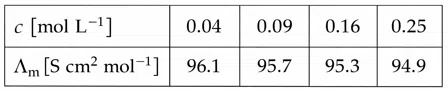
The value of constant A based on the above data [in S $$cm^{2}mol^{-1}/(mol/L)^{1/2}$$]unit is_______.
correct answer:- 4
Question 13
Given below are two statements : Statement (I) : Corrosion is an electrochemical phenomenon in which pure metal acts as an anode and impure metal as a cathode. Statement (II) : The rate of corrosion is more in alkaline medium than in acidic medium. In the light of the above statements, choose the correct answer from the options given below :
correct answer:- 3
Question 14
Which of the following electrolyte can be used to obtain $$H_2 S_2 O_8 $$ by the process of electrolysis?
correct answer:- 4
Question 15
A solution of aluminium chloride is electrolysed for 30 minutes using a current of 2 A . The amount of the aluminium deposited at the cathode is [Given : molar mass of aluminium and chlorine are 27g $$mol^{-1} $$ and 35.5 g $$mol^{-1}$$ respectively. Faraday constant $$=96500C mol^{-1}$$ ]
correct answer:- 2
Question 16
Standard electrode potentials for a few half cells are mentioned below :
$$E_{Cu^{2+}/Cu}^{\circ}=0.34 V,E_{Zn^{2+}/Zn}^{\circ}=-0.76 V\\E_{Ag^{+}/Ag}^{\circ}=0.80 V,E_{Mg^{2+}/Mg}^{\circ}=-2.37 V$$ Which one of the following cells gives the most negative value of $$\Delta G^{\circ}$$ ?
correct answer:- 1
Question 17
For the given cell $$Fe^{2+}_{(aq)} + Ag^+_{(aq)} \rightarrow Fe^{3+}_{(aq)} + Ag_{(s)},$$ the standard cell potential of the above reaction is Given: $$\begin{aligned}Ag^+ + e^- &\rightarrow Ag \qquad E^\circ = x\,V \\Fe^{2+} + 2e^- &\rightarrow Fe \qquad E^\circ = y\,V \\Fe^{3+} + 3e^- &\rightarrow Fe \qquad E^\circ = z\,V \end{aligned}$$
correct answer:- 3
Question 18
Based on the data given below: $$\begin{aligned}E^\circ_{\text{Cr}_2\text{O}_7^{2-}/\text{Cr}^{3+}} &= 1.33 \text{V} &E;^\circ_{\text{Cl}_2/\text{Cl}^-} &= 1.36\ \text{V} \\E^\circ_{\text{MnO}_4^-/\text{Mn}^{2+}} &= 1.51 \text{V} &E;^\circ_{\text{Cr}^{3+}/\text{Cr}} &= -0.74\ \text{V}\end{aligned}$$ the strongest reducing agent is:
correct answer:- 1
Question 19
Given below is the plot of the molar conductivity vs $$\sqrt{concentration}$$ for KCl in aqueous solution.
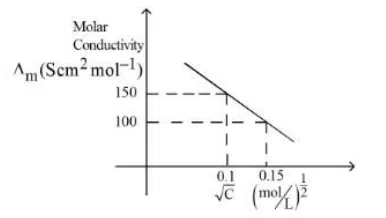
If, for the higher concentration of KCl solution, the resistance of the conductivity cell is 100Ω, then the resistance of the same cell with the dilute solution is $$'n'$$ Ω The value of $$x$$ is __________ (Nearest integer)
correct answer:- 150
Question 20
The molar conductivity of a weak electrolyte when plotted against the square root of its concentration, which of the following is expected to be observed ?
correct answer:- 2
Question 21
The standard reduction potential values of some of the p-block ions are given below. Predict the one with the strongest oxidising capacity.
correct answer:- 1
Question 22
For a $$Mg|Mg^{2+}(aq)||Ag^{+}(aq)|Ag$$ the correct Nernst Equation is :
correct answer:- 2
Question 23
$$FeO_{4}^{2-}\xrightarrow{+2.0_{V}} Fe^{3+}\xrightarrow{0.8_{V}} Fe^{2+}\xrightarrow{-0.5_{V}}Fe^{0}$$ In the above diagram, the standard electrode potentials are given in volts (over the arrow). The value of $$E_{FeO_{4}^{2-}/Fe^{2+}}^{0}$$ is
correct answer:- 2
Question 24
$$O_{2}$$ gas will be evolved as a product of electrolysis of : (A) an aqueous solution of $$AgNO_{3}$$ using silver electrodes. (B) an aqueous solution of $$AgNO_{3}$$ using platinum electrodes. (C) a dilute solution of $$H_{2}SO_{4}$$ using platinum electrodes. (D) a high concentration solution of $$H_{2}SO_{4}$$ using platinum electrodes. Choose the correct answer from the options given below :
correct answer:- 2
Question 25
Match List - I with List - II :

Choose the correct answer from the options given below :
correct answer:- 3
Question 26
What pressure (bar) of H₂ would be required to make emf of hydrogen electrode zero in pure water at 25°C?
correct answer:- 4
Question 27
One of the commonly used electrode is calomel electrode. Under which of the following categories, calomel electrode comes?
correct answer:- 4
Question 28
Electrolysis of 600 mL aqueous solution of NaCl for 5 min changes the pH of the solution to 12 . The current in Amperes used for the given electrolysis is ____. (Nearest integer).
correct answer:- 2
Question 29
The potential for the given half cell at 298K is $$- x \times 10^{-2}$$ V.
$$2H^+_{(aq)} + 2e^- \rightarrow H_2(g)$$, $$[H^+] = 1$$ M, $$P_{H_2} = 2$$ atm.
(Given $$2.303\ RT/F = 0.06$$ V, $$\log 2 = 0.3$$). The value of $$x$$ is:
correct answer:- 1
Question 30
The amount of electricity in Coulomb required for the oxidation of 1 mol of $$H_2O$$ to $$O_2$$ is ______ $$\times 10^5$$ C.
correct answer:- 2
Question 31
Consider the following redox reaction: $$MnO_4^- + H^+ + H_2C_2O_4 \rightleftharpoons Mn^{2+} + H_2O + CO_2$$
The standard reduction potentials are given as below:
$$E^0_{MnO_4^-/Mn^{2+}} = +1.51$$ V; $$E^0_{CO_2/H_2C_2O_4} = -0.49$$ V
If the equilibrium constant of the above reaction is given as $$K_{eq} = 10^x$$, then the value of $$x$$ = ______ (nearest integer)
correct answer:- 338
Question 32
The mass of silver (Molar mass of Ag : $$108 \text{ g mol}^{-1}$$) displaced by a quantity of electricity which displaces $$5600$$ mL of $$O_2$$ at S.T.P. will be ______ g.
correct answer:- 108
Question 33
Which of the following statements is not correct about rusting of iron?
correct answer:- 1
Question 34
The hydrogen electrode is dipped in a solution of pH = 3 at 25°C. The potential of the electrode will be $$-$$ _____ $$\times 10^{-2}$$ V.
$$\frac{2.303RT}{F} = 0.059$$ V. Round off the answer to the nearest integer.
correct answer:- 18
Question 35
The mass of zinc produced by the electrolysis of zinc sulphate solution with a steady current of $$0.015$$ A for $$15$$ minutes is _______ $$\times 10^{-4}$$ g. (Atomic mass of zinc $$= 65.4$$ amu)
correct answer:- 46
Question 36
A constant current was passed through a solution of $$AuCl_4^-$$ ion between gold electrodes. After a period of $$10.0$$ minutes, the increase in mass of cathode was $$1.314$$ g. The total charge passed through the solution is ______ $$\times 10^{-2}$$ F. (Given atomic mass of Au = 197)
correct answer:- 2
Question 37
The metals that are employed in the battery industries are A. Fe, B. Mn, C. Ni, D. Cr, E. Cd. Choose the correct answer from the options given below:
correct answer:- 1
Question 38
Identify the factor from the following that does not affect electrolytic conductance of a solution.
correct answer:- 2
Question 39
One Faraday of electricity liberates $$x \times 10^{-1}$$ gram atom of copper from copper sulphate, $$x$$ is ______.
correct answer:- 5
Question 40
The values of conductivity of some materials at 298.15 K in S m$$^{-1}$$ are $$2.1 \times 10^3$$, $$1.0 \times 10^{-16}$$, $$1.2 \times 10$$, $$3.91$$, $$1.5 \times 10^{-2}$$, $$1 \times 10^{-7}$$, $$1.0 \times 10^3$$. The number of conductors among the materials is
correct answer:- 4
Question 41
For a strong electrolyte, a plot of molar conductivity against (concentration)$$^{1/2}$$ is a straight line, with a negative slope, the correct unit for the slope is:
correct answer:- 2
Question 42
Fuel cell, using hydrogen and oxygen as fuels: A. has been used in spaceship, B. has an efficiency of 40% to produce electricity, C. uses aluminum as catalysts, D. is eco-friendly, E. is actually a type of Galvanic cell only. Choose the correct answer from the options given below:
correct answer:- 2
Question 43
The quantity of silver deposited when one coulomb charge is passed through $$AgNO_3$$ solution :
correct answer:- 2
Question 44
For the electrochemical cell, if $$E^0_{(M^{2+}/M)} = 0.46$$ V and $$E^0_{(X/X^{2-})} = 0.34$$ V. Which of the following is correct?
correct answer:- 4
Question 45
The reaction at cathode in the cells commonly used in clocks involves.
correct answer:- 2
Question 46
Molar ionic conductivities of divalent cation and anion are $$57 \text{ S cm}^2 \text{ mol}^{-1}$$ and $$73 \text{ S cm}^2 \text{ mol}^{-1}$$ respectively. The molar conductivity of solution of an electrolyte with the above cation and anion will be :
correct answer:- 3
Question 47
A conductivity cell with two electrodes (dark side) are half filled with infinitely dilute aqueous solution of a weak electrolyte. If volume is doubled by adding more water at constant temperature, the molar conductivity of the cell will -
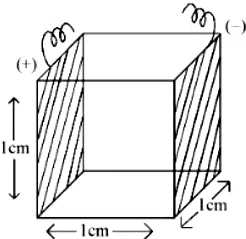
correct answer:- 3
Question 48
How can an electrochemical cell be converted into an electrolytic cell?
correct answer:- 3
Question 49
The emf of cell
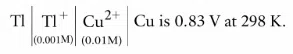
It could be increased by :
correct answer:- 2
Question 50
The reaction; $$\frac{1}{2}H_{2(g)} + AgCl_{(s)} \rightarrow H^+_{(aq)} + Cl^-_{(aq)} + Ag_{(s)}$$ occurs in which of the following galvanic cell :
correct answer:- 3

 Group
Group






.webp)














