Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The mass of silver (Molar mass of Ag : $$108 \text{ g mol}^{-1}$$) displaced by a quantity of electricity which displaces $$5600$$ mL of $$O_2$$ at S.T.P. will be ______ g.
Correct Answer: 108
At STP, 5600 mL of $$O_2$$ = 5.6 L of $$O_2$$.
Moles of $$O_2 = \frac{5.6}{22.4} = 0.25$$ mol.
The electrolytic reaction for oxygen at the anode:
$$2H_2O \to O_2 + 4H^+ + 4e^-$$
So 1 mol of $$O_2$$ requires 4 mol of electrons. Therefore, 0.25 mol $$O_2$$ requires $$0.25 \times 4 = 1$$ mol of electrons.
For silver deposition at the cathode:
$$Ag^+ + e^- \to Ag$$
1 mol of electrons deposits 1 mol of Ag = 108 g.
Mass of silver displaced = $$1 \times 108 = 108$$ g.
The answer is $$\boxed{108}$$ g.
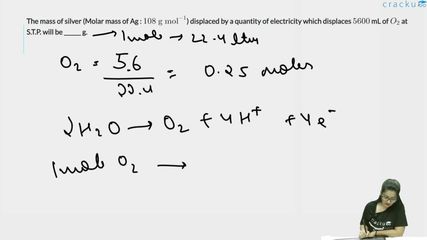
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
