JEE Chemical Thermodynamics PYQs
Another significant subject in the JEE Chemistry syllabus is JEE Chemical Thermodynamics PYQs. They are used to make you aware of the type of questions posed on this chapter and how familiar you are with the key concepts as system and surroundings, internal energy, enthalpy, entropy, Gibbs free energy, heat of reaction and spontaneity.
During the exam, the questions of chemical thermodynamics are normally considered in direct numerical form or concept-based questions. The advantage is that this chapter is easy to understand when you have your basics straight. When you have mastered the concepts and you know the formula to apply, it will be easier to answer the questions. Chemical thermodynamics is not something that you have to consider as a very hard chapter. It can be one of the more scoring portions to JEE Chemistry with revision and practice.
This blog will contain a simple formula PDF, a section containing key JEE Chemical Thermodynamics PYQs in the download form, a few questions with answers and a few questions to be solved independently. You will also come to know about some of the most common mistakes which the students usually commit and some of the few easy tips that can help you to save time during exam.
JEE Chemical Thermodynamics Important PYQs PDF
The most significant past year questions in chemical thermodynamics can be incorporated in this PDF. It can include such topics as system and surroundings, thermodynamic processes, internal energy, enthalpy change, Hess law, entropy, Gibbs free energy, spontaneity and heat of reaction.
These questions will be useful in practice to know more about the exam pattern. It will also make you faster, more accurate, and confident in case of the exam.
Important Formulas for JEE Chemical Thermodynamics PYQs
You only need a few important formulas and ideas to solve most chemical thermodynamics questions in JEE. These formulas help you understand heat changes, energy changes, spontaneity, and reaction behaviour more clearly.
You can download the full formula PDF from the link above. Here is a quick look at some of the main formulas:
Concept | Formula |
First Law of Thermodynamics | ΔU = q + w |
Work Done | w = -PΔV |
Enthalpy Change | ΔH = ΔU + PΔV |
Relation at Constant Pressure | qₚ = ΔH |
Relation at Constant Volume | qᵥ = ΔU |
Gibbs Free Energy | ΔG = ΔH - TΔS |
Entropy Change | ΔS = q rev / T |
Heat Capacity | C = q / ΔT |
Hess’s Law | Total enthalpy change is the same for all paths |
Spontaneous Process Condition | ΔG < 0 |
These formulas are commonly used in questions based on enthalpy, internal energy, entropy, free energy, spontaneity, and thermochemical equations. If you revise them properly, many JEE questions start to feel much easier.
Top 5 Common Mistakes to Avoid in JEE Chemical Thermodynamics PYQs
Many students find chemical thermodynamics confusing at first because it includes both formulas and concepts. But most mistakes happen because small details are missed while solving. Here are some common mistakes you should avoid:
Mixing up internal energy and enthalpy
Internal energy and enthalpy are related, but they are not the same. Internal energy is the total energy of the system, while enthalpy is mainly used when the process happens at constant pressure.
Using the wrong sign convention
In thermodynamics, signs matter a lot. Heat absorbed, heat released, work done by the system, and work done on the system must be handled carefully.
Confusing exothermic and endothermic processes
In an exothermic process, heat is released and ΔH is negative. In an endothermic process, heat is absorbed and ΔH is positive. Many students mix these up.
Forgetting the condition for spontaneity
A process is spontaneous when Gibbs free energy change is negative. Some students remember the formula but forget how to apply it in different cases.
Ignoring units
Always use proper units like joule, kilojoule, kelvin, and mole. Even if the method is correct, a unit mistake can change the final answer.
List of JEE Chemical Thermodynamics PYQs
Here is a short set of JEE-style chemical thermodynamics questions for practice. These include common question types from enthalpy, internal energy, free energy, and heat changes. Solving them regularly can help you become faster and more confident.
Question 1
Consider the following data :
$$\Delta_f H^\ominus$$ (methane, g) = - X kJ $$mol^{-1}$$
Enthalpy of sublimation of graphite = Y kJ $$mol^{-1}$$
Dissociation enthalpy of $$H_{2}$$ = Zkj $$mol^{-1}$$
The bond enthalpy of C - H bond is given by :
correct answer:- 1
Question 2
Which of the following graphs between pressure 'p' versus volume 'V' represents the maximum work done?
correct answer:- 2
Question 3
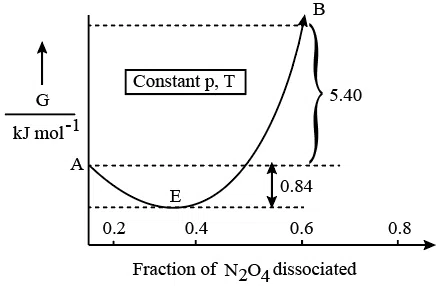
For the reaction $$N_{2}O_{4}\rightleftharpoons2NO_{2}$$ , graph is plotted as shown below. Identify correct statements.
A. Standard free energy change for the reaction is $$-5.40kJmol^{-1}$$.
B. As $$\triangle G^{\ominus}$$ in graph is positive, $$N_{2}O_{4}$$ will not dissociate into $$NO_{2}$$ at all.
C. Reverse reaction will go to completion.
D. When 1 mole of $$N_{2}O_{4}$$ changes into equilibrium mixture, value of $$\triangle G^{\ominus}$$ = -0.84kJ $$mol^{-1}$$.
E. When 2 mole of $$NO_{2}$$ changes into equilibrium mixture, $$\triangle G^{\ominus}$$ for equilibrium mixture is -6.24kJ $$mol^{-1}$$.
Choose the correct answer from the options given below:
correct answer:- 1
Question 4
Use the following data :
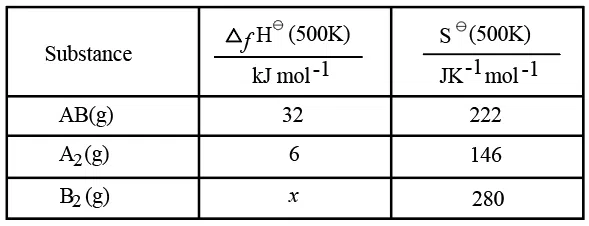
One mole each of $$A_{2}(g)$$ and $$B_{2}(g)$$ are taken in a 1 L closed flask and allowed to establish the equilibrium at 500K
$$A_{2}(g)+B_{2}(g)\rightleftharpoons2AB(g)$$
The value of x $$(in kJ mol^{-1})$$ is ____ . (Nearest integer)
(Given: log K=2.2 R= 8.3 kJ $$K^{-1} mol^{-1}$$)
correct answer:- 70
Question 5
If the enthalpy of sublimation of Li is 155 kj $$mol^{-1}$$, enthalpy of dissociation of $$F_{2}$$ is 150 kj $$mol^{-1}$$, ionization enthalpy of Li is 520 kj $$mol^{-1}$$, electron gain enthalpy of F is - 313 kj $$mol^{-1}$$, standard enthalpy of formation of LiF is - 594 kj $$mol^{-1}$$. The magnitude of lattice enthalpy of LiF is _________ kJ $$mol_{-1}$$. (Nearest Integer)
correct answer:- 1031
Question 6
A cup of water at 5°C (system) is placed in a microwave oven and the oven is turned on for one minute during which the water begins to boil. Which of the following option is true ?
correct answer:- 3
Question 7
Match the LIST-I with LIST-II

Choose the correct answer from the options given below:
correct answer:- 2
Question 8
Match the LIST-I with LIST-II
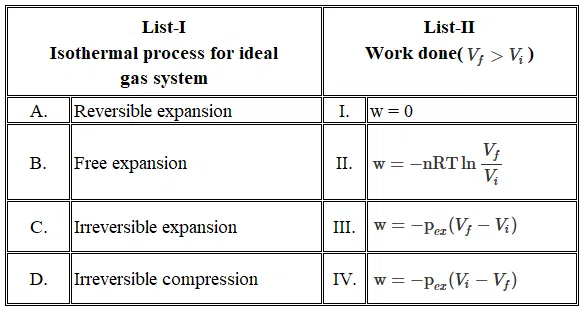
Choose the correct answer from the options given befow:
correct answer:- 2
Question 9
The heat of atomisation of methane and ethane are 'x' kJ $$mol^{-1}$$ and 'y' kJ $$mol^{-1}$$ respectively. The longest wavelength ($$\lambda$$) of light capable of breaking the C-C bond
can be expressed in SI unit as:
correct answer:- 3
Question 10
$$20.0 dm^{3}$$ of an ideal gas 'X' at 600 K and 0.5 MPa undergoes isothermal reversible expansion until pressme of the gas is 0.2 MPa. Which of the following option is correct?
(Given: $$\log 2 = 0.30 10 and \log 5 = 0.6989$$)
correct answer:- 4
Question 11

Match List - I with List - II.
Choose the correct answer from the options given below :
correct answer:- 3
Question 12
Consider the following cases of standard enthalpy of reaction ($$\Delta H_{r}^{\circ}$$ in kJ $$mol^{-1}$$)
$$C_{2}H_{6}(g)+\frac{7}{2}O_{2}(g) \rightarrow 2CO_{2}(g)+3H_{2}O(1)\Delta H_{1}^{\circ}=-1550$$
$$C(graphite)+O_{2}(g)\rightarrow CO_{2}(g)$$ $$\Delta H_{2}^{\circ}=-393.5$$ The magnitude of $$\Delta H_{fC_{2}H_{6}(g)}^{\circ}$$ is_______
$$H_{2}(g)+\frac{1}{2}O_{2}(g)\rightarrow H_{2}O(1)$$ $$\Delta H_{3}^{\circ}=-286$$
$$kJ mol^{-1}$$ (Nearest integer).
correct answer:- 95
Question 13
A liquid when kept inside a thermally insulated closed vessel $$25^{o}C$$ at was mechanically stirred from outside. What will be the correct option for the following thermodynamic parameters ?
correct answer:- 3
Question 14
The effect of temperature on spontaneity of reactions are represented as :

correct answer:- 1
Question 15
The bond dissociation enthalpy of $$X_{2}\Delta H_{bond}$$ calculated from the given data is_____$$kJmol^{-1}$$.(Nearest integer)
$$M^{+}X^{-}(s)\rightarrow M^{+}(g)+X^{-}(g)\Delta H_{lattice}^{*}=800kJmol^{-1}\\M(s)\rightarrow M(g)\Delta H_{sub}^{\circ}=100kJmol^{-1}\\M(g)\rightarrow M^{+}(g)+e^{-}(g)\Delta H_{i}=500kJmol^{-1}X(g)+e^{-}(g)\rightarrow X^{-}(g)\Delta H_{eg}^{*}=-300kJmol^{-1}\\M(g)+\frac{1}{2}X_{2}(g)\rightarrow M^{+}X^{-}(s)\Delta H_{f}^{\circ}=-400kJmol^{-1}$$
[Given : $$M^{+}X^{-}$$ is a pure ionic compound and X forms a diatomic molecule $$X_{2}$$ in gaseous state]
correct answer:- 200
Question 16
Let us consider an endothermic reaction which is non-spontaneous at the freezing point of water. However, the reaction is spontaneous at boiling point of water. Choose the correct option.
correct answer:- 4
Question 17
Standard entropies of $$X_2,\ Y_2$$ and $$XY_5$$ are $$70,\ 50$$ and $$110\,J\,K^{-1}mol^{-1}$$ respectively. The temperature in Kelvin at which the reaction $$\frac{1}{2}X_2 + \frac{5}{2}Y_2 \rightleftharpoons XY_5 \Delta H^\ominus = -35\,kJ\,mol^{-1}$$ will be at equilibrium is $$\underline{\hspace{2cm}}$$ (Nearest integer).}
correct answer:- 700
Question 18
Which of the following mixing of 1 M base and 1 M acid leads to the largest increase in temperature?
correct answer:- 3
Question 19
$$S(g) + \frac{3}{2}O_2(g) \rightarrow SO_3(g) + 2x\,\text{kcal} \\SO_2(g) + \frac{1}{2}O_2(g) \rightarrow SO_3(g) + y\,\text{kcal}\\\text{The heat of formation of } SO_2(g) \text{ is given by:}$$
correct answer:- 2
Question 20
Ice and water are placed in a closed container at a pressure of 1 atm and temperature 273.15 K . If pressure of the system is increased 2 times, keeping temperature constant, then identify correct observation from following
correct answer:- 2
Question 21
The formation enthalpies, $$\Delta H_{f}\ominus$$ for $$H_{g}$$ and $$O_{g}$$ are 220.0 and $$250.0 kJ mol^{-1}$$, respectively, at 298.15 K , and $$\Delta H_{f}\ominus$$ for $$H_{2}O_{g}$$ is $$-242.5 kJ mol^{-1}$$ at the same temperature. The average bond enthalpy of the $$O-H$$ bond in water at 298.15 K is __________$$kJ mol^{-1}$$(nearest integer).
correct answer:- 466
Question 22
J of energy is transferred as heat to 0.5 mol of Argon gas at 298 K and 1.00 atm. The final temperature and the change in internal energy respectively are: Given : $$R = 8.3 J K^{-1}mol^{-1}$$
correct answer:- 3
Question 23
Ice at $$-5^{\circ}C$$ is heated to become vapor with temperature of $$110^{\circ}C$$ at atmospheric pressure. The entropy change associated with this process can be obtained from
correct answer:- 2
Question 24
The standard enthalpy and standard entropy of decomposition of $$N_{2}O_{4}$$ to $$NO_{2}$$ are 55.0 kJ $$mol^{-1}$$ and 175.0 J/K/mol respectively. The standard free energy change for this reaction at $$25^{\circ}C$$ in J $$mol^{-1}$$ is ______ (Nearest integer)
correct answer:- 2850
Question 25
If $$C(\text{diamond}) \rightarrow C(\text{graphite}) + X\,\text{kJ mol}^{-1} C(\text{diamond}) + O_2(g) \rightarrow CO_2(g) + Y\,\text{kJ mol}^{-1}C(\text{graphite}) + O_2(g) \rightarrow CO_2(g) + Z\,\text{kJ mol}^{-1}$$ at constant temperature. Then
correct answer:- 4
Question 26
The enthalpy of formation of ethane (C₂H₆) from ethylene by addition of hydrogen where the bond-energies of C−H, C−C, C=C, H−H are 414 kJ, 347 kJ, 615 kJ and 435 kJ respectively is ______ kJ.
correct answer:- 125
Question 27
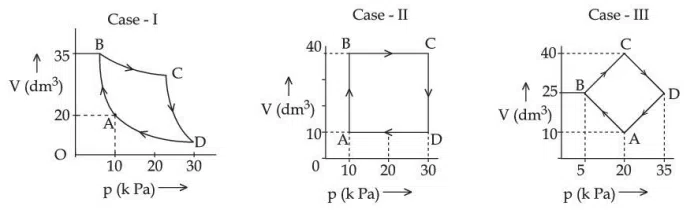
An ideal gas undergoes a cyclic transformation starting from the point A and coming back to the same point by tracing the path $$ A \rightarrow B \rightarrow C \rightarrow D \rightarrow A$$ as shown in the three cases above. Choose the correct option regarding $$\Delta U$$ :
correct answer:- 1
Question 28
Consider the following data : Heat of formation of $$CO_{2}$$(g)= -393.5 kJ $$mol^{-1}$$ Heat of formation of $$H_{2}0$$(l) = - 286.0 kJ $$mol^{-1}$$ Heat of combustion of benzene =-3267.0 kJ $$mol^{-1}$$ The heat of formation of benzene is _ . (Nearest integer)
correct answer:- 48
Question 29
Choose the correct option for free expansion of an ideal gas under adiabatic condition from the following :
correct answer:- 4
Question 30
For a certain reaction at 300 K, K = 10, then $$\Delta G°$$ for the same reaction is - ______ $$\times$$ $$10^{-1}$$ $$kJ mol^{-1}$$. (Given $$R = 8.314 \text{ J K}^{-1} \text{mol}^{-1}$$)
correct answer:- 57
Question 31
If three moles of an ideal gas at $$300$$ K expand isothermally from $$30 \text{ dm}^3$$ to $$45 \text{ dm}^3$$ against a constant opposing pressure of $$80$$ kPa, then the amount of heat transferred is _________ J.
correct answer:- 1200
Question 32
For a certain thermochemical reaction $$M \rightarrow N$$ at $$T = 400$$ K, $$\Delta H^o = 77.2$$ kJ mol$$^{-1}$$, $$\Delta S^o = 122$$ J K$$^{-1}$$, log equilibrium constant ($$\log K$$) is $$-$$ _____ $$\times 10^{-1}$$.
correct answer:- 37
Question 33
Which of the following is not correct?
correct answer:- 2
Question 34
Standard enthalpy of vapourisation for $$CCl_4$$ is $$30.5$$ kJ mol$$^{-1}$$. Heat required for vapourisation of $$284$$ g of $$CCl_4$$ at constant temperature is ______ kJ. (Given molar mass in g mol$$^{-1}$$; C = 12, Cl = 35.5)
correct answer:- 56
Question 35
An ideal gas undergoes a cyclic transformation starting from the point $$A$$ and coming back to the same point by tracing the path $$A \rightarrow B \rightarrow C \rightarrow A$$ as shown in the diagram. The total work done in the process is _____ J.
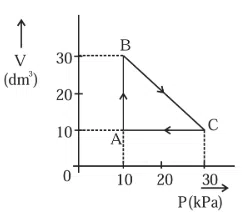
correct answer:- 200
Question 36
Two reactions are given below:
$$2Fe_{(s)} + \frac{3}{2}O_{2(g)} \rightarrow Fe_2O_{3(s)}$$, $$\Delta H° = -822$$ kJ/mol
$$C_{(s)} + \frac{1}{2}O_{2(g)} \rightarrow CO_{(g)}$$, $$\Delta H° = -110$$ kJ/mol
Then enthalpy change for following reaction:
$$3C_{(s)} + Fe_2O_{3(s)} \rightarrow 2Fe_{(s)} + 3CO_{(g)}$$
correct answer:- 492
Question 37
Consider the following reaction at 298 K. $$\frac{3}{2}O_{2(g)} \rightleftharpoons O_{3(g)}$$, $$K_p = 2.47 \times 10^{-29}$$. $$\Delta_r G^0$$ for the reaction is _________ kJ. (Given $$R = 8.314 \text{ JK}^{-1}\text{mol}^{-1}$$) Round off your answer to the nearest integer.
correct answer:- 163
Question 38
If 5 moles of an ideal gas expands from 10 L to a volume of 100 L at 300 K under isothermal and reversible condition then work, $$w$$, is $$-x$$ J. The value of $$x$$ is (Given $$R = 8.314$$ J K$$^{-1}$$ mol$$^{-1}$$)
correct answer:- -28721
Question 39
Three moles of an ideal gas are compressed isothermally from $$60$$ L to $$20$$ L using constant pressure of $$5$$ atm. Heat exchange $$Q$$ for the compression is $$-$$ ______ Lit. atm.
correct answer:- 200
Question 40
Combustion of 1 mole of benzene is expressed at $$C_6H_6(l) + \frac{15}{2}O_2(g) \rightarrow 6CO_2(g) + 3H_2O(l)$$. The standard enthalpy of combustion of 2 mol of benzene is $$-'x'$$ kJ. $$x =$$ ______ Given: 1. Standard enthalpy of formation of 1 mol of $$C_6H_6(l)$$, for the reaction $$6C \text{(graphite)} + 3H_2(g) \rightarrow C_6H_6(l)$$ is $$48.5 \text{ kJ mol}^{-1}$$. 2. Standard enthalpy of formation of 1 mol of $$CO_2(g)$$, for the reaction $$C \text{(graphite)} + O_2(g) \rightarrow CO_2(g)$$ is $$-393.5 \text{ kJ mol}^{-1}$$. 3. Standard enthalpy of formation of 1 mol of $$H_2O(l)$$, for the reaction $$H_2(g) + \frac{1}{2}O_2(g) \rightarrow H_2O(l)$$ is $$-286 \text{ kJ mol}^{-1}$$.
correct answer:- 6535
Question 41
Which of the following gives a positive test with ninhydrin?
correct answer:- 2
Question 42
DNA molecule contains 4 bases whose structures are shown below. One of the structures is not correct, identify the incorrect base structure.
correct answer:- 2
Question 43
Identify the product (A) in the following reaction.
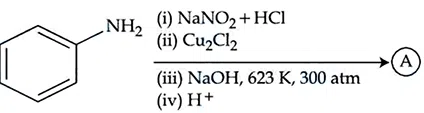
correct answer:- 4
Question 44
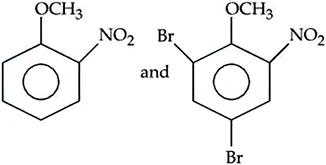
The incorrect statements regarding enzymes are : (A) Enzymes are biocatalysts. (B) Enzymes are non-specific and can catalyse different kinds of reactions. (C) Most Enzymes are globular proteins. (D) Enzyme - oxidase catalyses the hydrolysis of maltose into glucose. Choose the correct answer from the option given below :
correct answer:- 2
Question 45
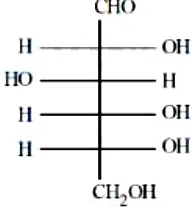
The incorrect statement regarding the given structure is
correct answer:- 1
Question 46
The total number of carbon atoms present in tyrosine, an amino acid, is _____
correct answer:- 9
Question 47
Total number of essential amino acid among the given list of amino acids is _______ Arginine, Phenylalanine, Aspartic acid, Cysteine, Histidine, Valine, Proline
correct answer:- 4
Question 48
When $$\Delta H_{vap} = 30 \text{ kJ/mol}$$ and $$\Delta S_{vap} = 75 \text{ J mol}^{-1} \text{K}^{-1}$$, then the temperature of vapour, at one atmosphere is ______ K.
correct answer:- 400
Question 49
At 25°C, the enthalpy of the following processes are given:
$$H_2(g) + O_2(g) \rightarrow 2OH(g) \quad \Delta H^o = 78 \text{ kJ mol}^{-1}$$
$$H_2(g) + \frac{1}{2}O_2(g) \rightarrow H_2O(g) \quad \Delta H^o = -242 \text{ kJ mol}^{-1}$$
$$H_2(g) \rightarrow 2H(g) \quad \Delta H^o = 436 \text{ kJ mol}^{-1}$$
$$\frac{1}{2}O_2(g) \rightarrow O(g) \quad \Delta H^o = 249 \text{ kJ mol}^{-1}$$
What would be the value of X for the following reaction? (Nearest integer)
$$H_2O(g) \rightarrow H(g) + OH(g) \quad \Delta H^o = X \text{ kJ mol}^{-1}$$
correct answer:- 499
Question 50
0.3 g of ethane undergoes combustion at $$27°C$$ in a bomb calorimeter. The temperature of calorimeter system (including the water) is found to rise by $$0.5°C$$. The heat evolved during combustion of ethane at constant pressure is ______ $$\text{kJmol}^{-1}$$.
(Nearest integer)
[Given : The heat capacity of the calorimeter system is $$20 \text{ kJ K}^{-1}$$, $$R = 8.3 \text{ JK}^{-1} \text{ mol}^{-1}$$. Atomic mass of C and H are 12 and 1 $$\text{g mol}^{-1}$$ respectively]
correct answer:- 1006

 Group
Group






.webp)












