Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
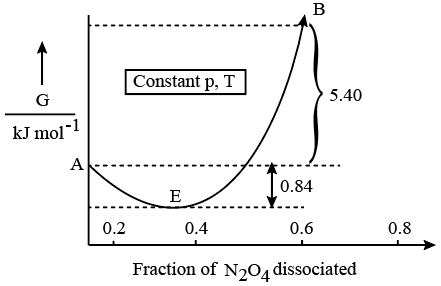
For the reaction $$N_{2}O_{4}\rightleftharpoons2NO_{2}$$ , graph is plotted as shown below. Identify correct statements.
A. Standard free energy change for the reaction is $$-5.40kJmol^{-1}$$.
B. As $$\triangle G^{\ominus}$$ in graph is positive, $$N_{2}O_{4}$$ will not dissociate into $$NO_{2}$$ at all.
C. Reverse reaction will go to completion.
D. When 1 mole of $$N_{2}O_{4}$$ changes into equilibrium mixture, value of $$\triangle G^{\ominus}$$ = -0.84kJ $$mol^{-1}$$.
E. When 2 mole of $$NO_{2}$$ changes into equilibrium mixture, $$\triangle G^{\ominus}$$ for equilibrium mixture is -6.24kJ $$mol^{-1}$$.
Choose the correct answer from the options given below:
We need to evaluate the given statements concerning the thermodynamic equilibrium of the dissociation of dinitrogen tetroxide ($$N_2O_4 \rightleftharpoons 2NO_2$$) based on standard free energy calculations.
Statement (A): Standard free energy change for the reaction is $$-5.40 \text{ kJ mol}^{-1}$$. Based on standard thermodynamic data at $$298 \text{ K}$$, the standard Gibbs free energy change ($$\Delta G^{\ominus}$$) for the dissociation of $$N_2O_4$$ into $$2NO_2$$ is approximately $$+5.40 \text{ kJ mol}^{-1}$$, which is positive, not negative. This is FALSE.
Statement (B): As $$\Delta G^{\ominus}$$ in graph is positive, $$N_2O_4$$ will not dissociate into $$NO_2$$ at all. A positive $$\Delta G^{\ominus}$$ signifies that the reactants are favored over products under standard state conditions ($$1 \text{ atm}$$). However, it does not mean the reaction will not proceed at all; it will still progress to a certain extent until dynamic chemical equilibrium is established. This is FALSE.
Statement (C): Reverse reaction will go to completion. Even though the reverse dimerization reaction is highly thermodynamically favored ($$\Delta G^{\ominus} = -5.40 \text{ kJ mol}^{-1}$$), it forms a dynamic chemical equilibrium mixture and will not go to absolute completion ($$100\%$$). This is FALSE.
Statement (D): When 1 mole of $$N_2O_4$$ changes into equilibrium mixture, value of $$\Delta G^{\ominus}$$ = -0.84kJ $$mol^{-1}$$. Looking closely at the free energy profile of the reaction mixture, the total change in free energy ($$\Delta G$$) when 1 mole of pure $$N_2O_4$$ transitions to the minimum point of the curve (equilibrium mixture) is $$-0.84 \text{ kJ}$$. This is TRUE.
Statement (E): When 2 mole of $$NO_2$$ changes into equilibrium mixture, $$\Delta G^{\ominus}$$ for equilibrium mixture is -6.24kJ $$mol^{-1}$$. Starting from 2 moles of pure $$NO_2$$ on the right side of the curve, the free energy change ($$\Delta G$$) dropped to reach the dynamic equilibrium minimum point corresponds to $$-5.40 \text{ kJ} + (-0.84 \text{ kJ}) = -6.24 \text{ kJ}$$. This is TRUE.
Statements (D) and (E) are correct.
Answer: Option A — D and E only

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
