Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the following reaction sequence
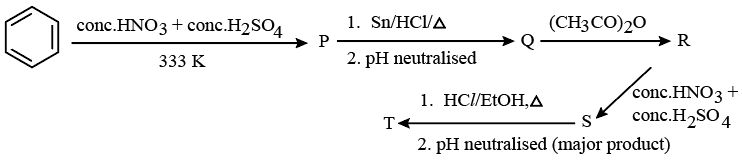
The percentage of nitrogen in product 'T' formed is ____ %. (Nearest integer)
(Given molar mass in g $$mol^{-1}$$ H : 1, C: 12, N : 14, 0: 16)
Correct Answer: 20
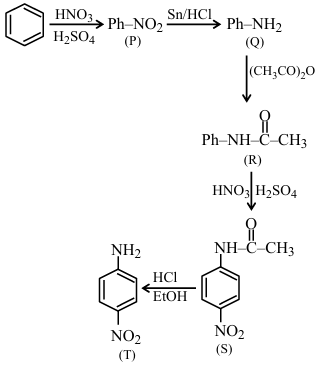
Mol. wt. = $$6\times\ 12+(6\times\ 1)+(2\times\ 14)+(2\times\ 16)=138$$
% N =$$\frac{\ 28}{138}\times\ 100$$ = 20.29 %
Nearest integer = 20%

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
