Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Pre-exponential factors of two different reactions of same order are identical. Let activation energy of first reaction exceeds the activation energy of second reaction by 20 kJ $$mol^{-1}$$. If $$k_{1}\text{ and }k_{2}$$ are the rate constants of first and second reaction respectively at 300 K, then In $$\frac{k_{2}}{k_{1}}$$ will be ___.
(nearest integer) $$[R=8.3JK^{-1}mol^{-1}]$$
Correct Answer: 8
We are given two reactions with the same pre-exponential factor ($$A$$) but different activation energies. We need to find $$\ln\frac{k_2}{k_1}$$.
$$E_{a1} - E_{a2} = 20 \text{ kJ mol}^{-1} = 20000 \text{ J mol}^{-1}$$
$$T = 300 \text{ K}$$
$$R = 8.3 \text{ J K}^{-1} \text{mol}^{-1}$$
Pre-exponential factors: $$A_1 = A_2 = A$$
The Arrhenius equation relates the rate constant to activation energy:
$$k = A \cdot e^{-E_a / RT}$$
For reaction 1: $$k_1 = A \cdot e^{-E_{a1}/RT}$$
For reaction 2: $$k_2 = A \cdot e^{-E_{a2}/RT}$$
Since both reactions have the same pre-exponential factor $$A$$:
$$\frac{k_2}{k_1} = \frac{A \cdot e^{-E_{a2}/RT}}{A \cdot e^{-E_{a1}/RT}} = e^{(E_{a1} - E_{a2})/RT}$$
$$\ln\frac{k_2}{k_1} = \frac{E_{a1} - E_{a2}}{RT}$$
$$\ln\frac{k_2}{k_1} = \frac{20000}{8.3 \times 300} = \frac{20000}{2490}$$
$$\ln\frac{k_2}{k_1} = 8.032 \approx 8$$
The answer is 8.
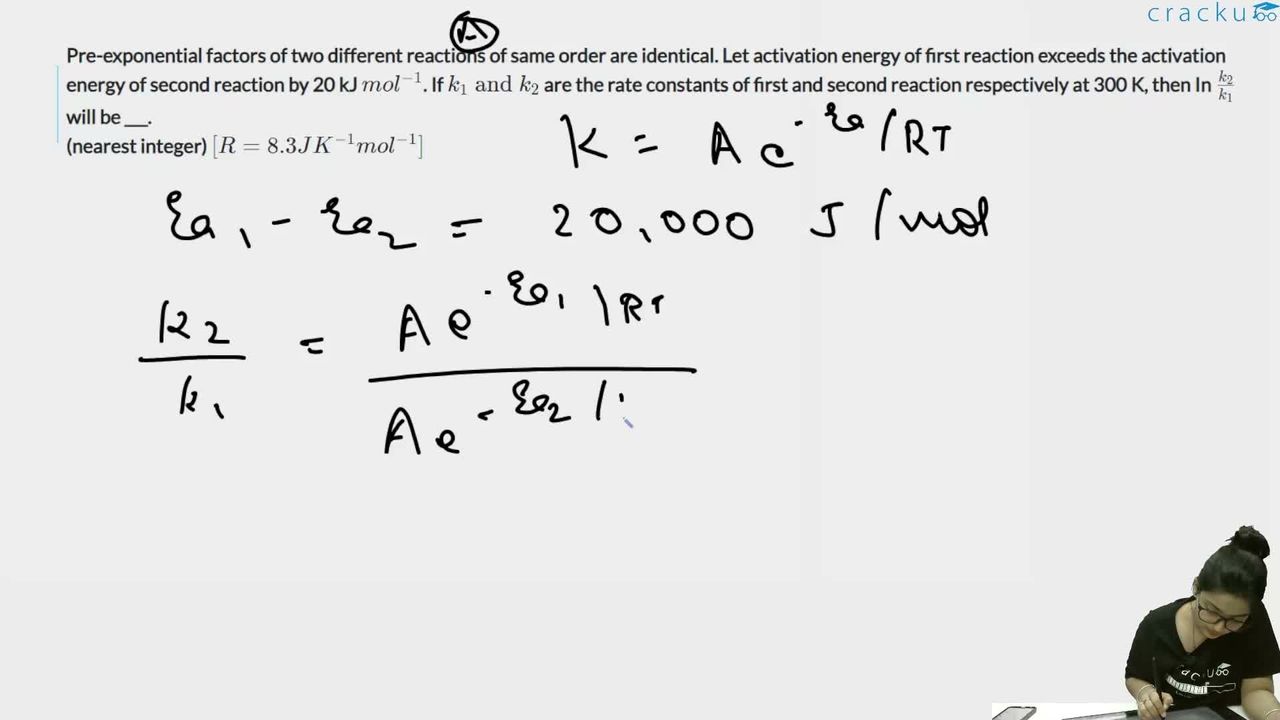
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
