Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Use the following data :
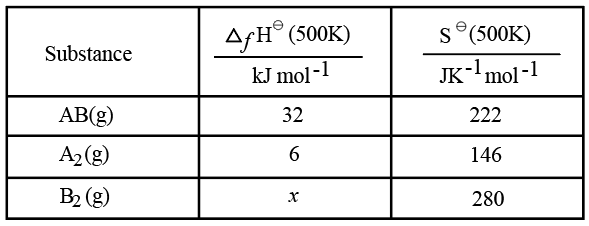
One mole each of $$A_{2}(g)$$ and $$B_{2}(g)$$ are taken in a 1 L closed flask and allowed to establish the equilibrium at 500K
$$A_{2}(g)+B_{2}(g)\rightleftharpoons2AB(g)$$
The value of x in $$( kJ mol^{-1})$$ is ____ . (Nearest integer)
(Given: log K=2.2 R= 8.3 kJ $$K^{-1} mol^{-1}$$)
Correct Answer: 70
One mole each of A₂(g) and B₂(g) are taken in a 1 L closed flask and allowed to establish equilibrium at 500 K:
A₂(g) + B₂(g) ⇌ 2AB(g)
Find the value of x (in kJ mol⁻¹).
(Given: log K = 2.2, R = 8.3 J K⁻¹ mol⁻¹)
ΔG° = ΔH° − TΔS°
ΔG° = −2.303 RT log K
ΔG° = −2.303 × R × T × log K
= −2.303 × 8.3 × 500 × 2.2
≈ −21000 J ≈ −21 kJ
ΔH° = [2 × ΔfH°(AB)] − [ΔfH°(A₂) + ΔfH°(B₂)]
= (2 × 32) − (6 + x)
= 64 − 6 − x
= 58 − x
ΔS° = [2 × S°(AB)] − [S°(A₂) + S°(B₂)]
= (2 × 222) − (146 + 280)
= 444 − 426
= 18 J K⁻¹
−21 = (58 − x) − (500 × 18 / 1000)
−21 = (58 − x) − 9
−21 = 49 − x
x = 70 kJ mol⁻¹
x = 70 kJ mol⁻¹

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
