Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
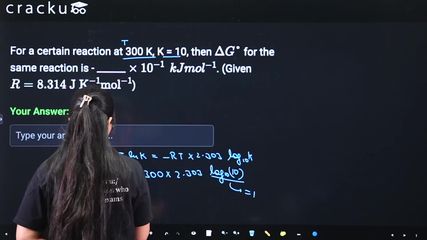
For a certain reaction at 300 K, K = 10, then $$\Delta G°$$ for the same reaction is - ______ $$\times$$ $$10^{-1}$$ $$kJ mol^{-1}$$. (Given $$R = 8.314 \text{ J K}^{-1} \text{mol}^{-1}$$)
Correct Answer: 57
$$\Delta G^\circ = -2.303 \cdot R \cdot T \cdot \log_{10}(K)$$
$$\Delta G^\circ = -2.303 \times 8.314 \times 300 \times 1$$
$$\Delta G^\circ = -5744.14 \text{ J mol}^{-1}$$
To convert Joules to kilojoules, divide by $$1000$$:
$$\Delta G^\circ = -5.74414 \text{ kJ mol}^{-1}$$
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
