JEE P-Block Elements PYQs
JEE P-Block Elements PYQs are an important part of the JEE Chemistry syllabus. They help you understand what kind of questions are asked from this chapter and how well you know the key concepts, such as group trends, electronic configuration, oxidation states, anomalous behaviour, chemical properties, important compounds, and uses of p-block elements.
In the exam, questions from p-block elements usually come as direct theory-based questions or concept-based problems. The good thing is that this chapter becomes much easier when your basics are clear. Once you understand the trends, reactions, and exceptions properly, solving questions becomes much more manageable. You do not need to think of p-block elements as a difficult chapter. With regular revision and smart practice, it can become one of the more scoring parts of JEE Chemistry.
In this blog, you will find a simple formula PDF, a section for important JEE P-Block Elements PYQs in download format, a few practice questions with answers, and some extra questions to solve on your own. You will also go through common mistakes students often make and a few simple tips to save time in the exam.
JEE P-Block Elements Important PYQs PDF
This PDF can include the most important previous year questions from p-block elements. It may cover topics like group 13 to group 18 elements, electronic configuration, oxidation states, inert pair effect, anomalous behaviour, acidic and basic nature, important compounds, and common chemical reactions.
Practicing these questions will help you understand the exam pattern better. It will also improve your speed, accuracy, and confidence before the exam.
Important Formulas for JEE P-Block Elements PYQs
You only need a few important rules, trends, and facts to solve most p-block elements questions in JEE. These help you understand oxidation states, acidity, stability, reactivity, and important compounds more clearly.
You can download the full formula PDF from the link above. Here is a quick look at some of the main rules and trends:
Concept | Formula / Rule |
General Valence Shell Configuration | ns² np¹ to ns² np⁶ |
Group 13 Configuration | ns² np¹ |
Group 14 Configuration | ns² np² |
Group 15 Configuration | ns² np³ |
Group 16 Configuration | ns² np⁴ |
Group 17 Configuration | ns² np⁵ |
Group 18 Configuration | ns² np⁶ |
Oxidation State Trend | Variable oxidation states are common in p-block elements |
Inert Pair Effect | Down the group, lower oxidation states become more stable |
Acidic Nature of Oxides | Increases from left to right across a period |
These rules and trends are often used in questions based on electronic configuration, oxidation states, periodic trends, important compounds, and chemical properties. If you revise them properly, many JEE questions will feel much easier to solve.
Top 5 Common Mistakes to Avoid in JEE P-Block Elements PYQs
Many students feel that p-block elements is hard because it includes a lot of facts and exceptions. But most mistakes happen because small details are missed while revising or solving. Here are some common mistakes you should avoid:
Mixing up group trends
Students often confuse trends like atomic size, ionization enthalpy, oxidation state, and acidic nature. It is important to revise these trends carefully, both group-wise and period-wise.
Forgetting anomalous behaviour
The first element of a group often behaves differently from the others. If you ignore these exceptions, you may end up choosing the wrong option.
Confusing oxidation states
P-block elements often show more than one oxidation state. Many students forget which oxidation state becomes more stable in heavier elements.
Skipping important compounds
Compounds like borax, boric acid, ammonia, nitric acid, ozone, sulphuric acid, bleaching powder, and xenon compounds are very important. Students sometimes leave them out and lose easy marks.
Ignoring reaction-based questions
This chapter is not only about trends and theory. Many questions are based on reactions, products, and chemical behaviour. Read each reaction carefully before answering.
List of JEE P-Block Elements PYQs
Here is a short set of JEE-style p-block elements questions for practice. These include common question types from group trends, oxidation states, compounds, and chemical properties. Solving them regularly can help you become faster and more confident.
Question 1
Given below are two statements:
Statement I: The number of pairs among $$[SiO_{2},CO_{2}],[SnO,SnO_{2}],[PbO,PbO_{2]}\text{ and } [GeO,GeO_{2}]$$, which contain oxides that are both amphoteric is 2.
Statement ll: $$BF_{3}$$ is an electron deficient molecule, can act as a Lewis add, forms adduct with $$NH_{3}$$ and has a trigonal planar geometry.
In the light of the above statements, choose the correct answer from the options given below:
correct answer:- 4
Question 2
Given below are two statements:
Statement I : Elements 'X' and 'Y' are the most and least electronegative elements, respectively among N, As, Sb and P. The nature of the oxides $$X_{2}O_{3}$$ and $$y_{2}O_{3}$$ is acidic and
amphoteric, respectively.
Statement II:$$BCl_{3}$$ is covalent in nature and gets hydrolysed in water. It produces $$[B(OH)_{4}]^{-}$$ and $$[B(H_{2}O)_{6}]^{3+}$$ in aqueous medium.
In the Light of the above statements, choose the correct answer from the options given below:
correct answer:- 4
Question 3
The correct statements from the following are:
A. Ionic radii of trivalent cations of group 13 elements decreases down the group.
B. Electronegativity of group 13 elements decreases down the group.
C. Among the group 13 elements, Boron has highest first ionisation enthalpy.
D. The trichloride and triiodide of group 13 elements are covalent in nature.
Choose the correct answer from the options given below :
correct answer:- 4
Question 4
Given below are two statements:
Statement I: The halogen that makes longest bond with hydrogen in HX, has the smallest covalent radius in its group.
Statement II: A group 15 element's hydride $$EH_{3}$$ has the lowest boiling point among corresponding hydrides of other group 15 elements. The maximum covalency of that element E is 4.
In the light of the above statements, choose the correct answer from the options given below
correct answer:- 4
Question 5
A 'p'-block element (E) and hydrogen form a binary cation $$(EH_{x})^{+}$$ , while $$EH_{3}$$ on treatment with $$K_{2}HgI_{4}$$ in alkaline medium gives a precipitate of basic mercury(II)amido- iodine. Given below are first ionisation enthalpy values ($$kJ mol^{-1}$$) for first element each from group 13, 14, 15 and 16. Identify the correct first ionisation enthalpy value for element E.
correct answer:- 4
Question 6
"X" is an oxoanion of the lightest element of group 7 (in the periodic table). The metal is in +6 oxidation state in "X". The color of the potassium salt of X is
correct answer:- 4
Question 7
Choose the INCORRECT statement
correct answer:- 3
Question 8
Regarding the hydrides of group 15 elements $$EH_{3}$$(E = N, P, As, Sb), select the correct statement from the following:
A. The stability of hydrides decreases down the group.
B. The basicity of hydrides decreases down the group.
C. The reducing character increases down the group.
D. The boiling point increases down the group.
Choose the correct answer from the options given below:
correct answer:- 1
Question 9
It is noticed that $$Pb^{2+}$$ is more stable than $$Pb^{4+}$$ but $$Sn^{2+}$$ is less stable than $$Sn^{4+}$$
Observe the following reactions.
$$ PbO_2+Pb\rightarrow 2PbO;\triangle_rG^{o}(1) $$
$$ SnO_2+Sn\rightarrow 2SnO;\triangle_rG^{o}(2) $$
Identify the correct set from the following
correct answer:- 2
Question 10
Consider the following reactions
$$\begin{aligned}\mathrm{Na_2B_4O_7} & \xrightarrow{\Delta} 2X + Y \\[6pt]\mathrm{CuSO_4} + Y &\xrightarrow{\text{Non-luminous flame}} Z + \mathrm{SO_3}\\[6pt]2Z + 2X + \mathrm{C}&\xrightarrow{\text{Luminous flame}} 2Q +\mathrm{Na_2B_4O_7} + \mathrm{CO}\end{aligned}$$
The oxidation states of Cu in Z and Q, respectively are:
correct answer:- 2
Question 11
The large difference between the melting and boiling points of oxygen and sulphur may be explained on the basis of
correct answer:- 1
Question 12
Given below are two statements :
Statement (I) : The first ionization energy of Pb is greater than that of Sn .
Statement (II) : The first ionization energy of Ge is greater than that of Si .
In the light of the above statements, choose the correct answer from the options given below :
correct answer:- 2
Question 13
The incorrect statement among the following is options .
correct answer:- 2
Question 14
The type of oxide formed by the element among Li, Na, Be, Mg, B and Al that has the least atomic radius is :
correct answer:- 2
Question 15
First ionisation enthalpy values of first four group 15 elements are given below. Choose the correct value for the element that is a main component of apatite family :
correct answer:- 3
Question 16
The number of the correct reaction(s) among the following is ______
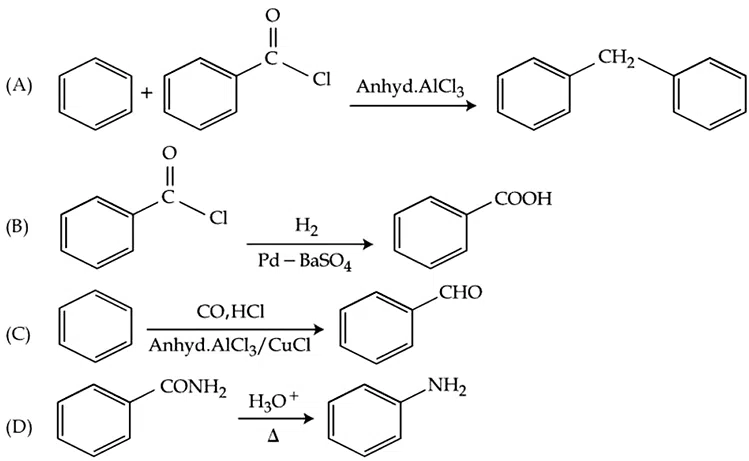
correct answer:- 1
Question 17
Identify the inorganic sulphides that are yellow in colour : (A) $$\mathrm{(NH_4)_2S}$$ (B) $$\mathrm{PbS}$$ (C) $$\mathrm{CuS}$$ (D) $$\mathrm{As_2S_3}$$ (E) $$\mathrm{As_2S_5}$$ Choose the correct answer from the options given below :
correct answer:- 1
Question 18
A group 15 element forms $$d\pi-d\pi$$ bond with transition metals. It also forms hydride, which is a strongest base among the hydrides of other group members that form $$d\pi-d\pi$$ bond. The atomic number of the element is ____.
correct answer:- 15
Question 19
Among the following oxides of p-block elements: $$Cl_2O_7$$, $$CO$$, $$PbO_2$$, $$N_2O$$, $$NO$$, $$Al_2O_3$$, $$SiO_2$$, $$N_2O_5$$, $$SnO_2$$, the number of amphoteric oxides is:
correct answer:- 3
Question 20
Given below are two statements:
Statement (I): $$SiO_2$$ and $$GeO_2$$ are acidic while SnO and PbO are amphoteric in nature.
Statement (II): Allotropic forms of carbon are due to property of catenation and $$p\pi - d\pi$$ bond formation.
In the light of the above statements, choose the most appropriate answer from the options given below:
correct answer:- 3
Question 21
The strongest reducing agent among the following is:
correct answer:- 3
Question 22
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Melting point of Boron (2453 K) is unusually high in group 13 elements.
Reason (R) : Solid Boron has very strong crystalline lattice.
In the light of the above statements, choose the most appropriate answer from the options given below:
correct answer:- 2
Question 23
Element not showing variable oxidation state is :
correct answer:- 4
Question 24
Given below are two statements:
Statement (I) : Oxygen being the first member of group 16 exhibits only -2 oxidation state.
Statement (II) : Down the group 16 stability of +4 oxidation state decreases and +6 oxidation state increases.
In the light of the above statements, choose the most appropriate answer from the options given below:
correct answer:- 3
Question 25
Match List-I with List-II.
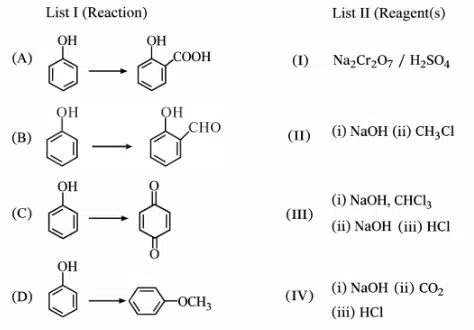
Choose the correct answer from the options given below:
correct answer:- 4
Question 26
Given below are two statements :
Statement I : The electronegativity of group 14 elements from Si to Pb gradually decreases.
Statement II : Group 14 contains non-metallic, metallic, as well as metalloid elements.
In the light of the above statements, choose the most appropriate from the options given below :
correct answer:- 1
Question 27
Identify the incorrect pair from the following :
correct answer:- 1
Question 28
Match List I with List II

Choose the correct answer from the options given below:
correct answer:- 4
Question 29
Anomalous behaviour of oxygen is due to its
correct answer:- 3
Question 30
Match List I with List II
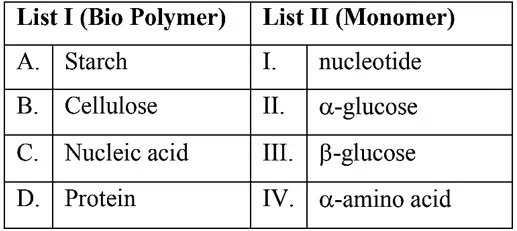
Choose the correct answer from the options given below:
correct answer:- 4
Question 31
Match List - I with List-II

correct answer:- 4
Question 32
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: $$H_2Te$$ is more acidic than $$H_2S$$.
Reason R: Bond dissociation enthalpy of $$H_2Te$$ is lower than $$H_2S$$.
In the light of the above statements, choose the most appropriate from the options given below.
correct answer:- 2
Question 33
Choose the correct statements about the hydrides of group 15 elements.
A. The stability of the hydrides decreases in the order $$NH_3 > PH_3 > AsH_3 > SbH_3 > BiH_3$$
B. The reducing ability of the hydrides increases in the order $$NH_3 < PH_3 < AsH_3 < SbH_3 < BiH_3$$
C. Among the hydrides, $$NH_3$$ is strong reducing agent while $$BiH_3$$ is mild reducing agent.
D. The basicity of the hydrides increases in the order $$NH_3 < PH_3 < AsH_3 < SbH_3 < BiH_3$$
Choose the most appropriate from the options given below:
correct answer:- 3
Question 34
Consider the oxides of group 14 elements $$SiO_2, GeO_2, SnO_2, PbO_2, CO$$ and $$GeO$$. The amphoteric oxides are
correct answer:- 3
Question 35
Give below are two statements:
Statement-I: Noble gases have very high boiling points.
Statement-II: Noble gases are monoatomic gases. They are held together by strong dispersion forces. Because of this they are liquefied at very low temperature. Hence, they have very high boiling points.
In the light of the above statements, choose the correct answer from the options given below:
correct answer:- 4
Question 36
Molar mass of the salt from $$NaBr, NaNO_3, KI$$ and $$CaF_2$$ which does not evolve coloured vapours on heating with concentrated $$H_2SO_4$$ is ____ g mol$$^{-1}$$. (Molar mass in g mol$$^{-1}$$: Na: 23, N: 14, K: 39, O: 16, Br: 80, I: 127, F: 19, Ca: 40)
correct answer:- 78
Question 37
Given below are two statements:
Statement I: Group 13 trivalent halides get easily hydrolysed by water due to their covalent nature.
Statement II: AlCl$$_3$$ upon hydrolysis in acidified aqueous solution forms octahedral $$[Al(H_2O)_6]^{3+}$$ ion.
In the light of the above statements, choose the correct answer from the options given below:
correct answer:- 4
Question 38
Choose the correct statements from the following:
A. All group 16 elements form oxides of general formula $$EO_2$$ and $$EO_3$$ where $$E = S, Se, Te$$ and $$Po$$. Both the types of oxides are acidic in nature.
B. $$TeO_2$$ is an oxidising agent while $$SO_2$$ is reducing in nature.
C. The reducing property decreases from $$H_2S$$ to $$H_2Te$$ down the group.
D. The ozone molecule contains five lone pairs of electrons.
Choose the correct answer from the options given below:
correct answer:- 4
Question 39
When $$MnO_2$$ and $$H_2SO_4$$ is added to a salt (A), the greenish yellow gas liberated as salt (A) is:
correct answer:- 4
Question 40
The correct statements from the following are : (A) The decreasing order of atomic radii of group 13 elements is $$Tl > In > Ga > Al > B$$. (B) Down the group 13 electronegativity decreases from top to bottom. (C) Al dissolves in dil. HCl and liberates $$H_2$$ but conc. $$HNO_3$$ renders Al passive by forming a protective oxide layer on the surface. (D) All elements of group 13 exhibits highly stable +1 oxidation state. (E) Hybridisation of Al in $$[Al(H_2O)_6]^{3+}$$ ion is $$sp^3d^2$$. Choose the correct answer from the options given below :
correct answer:- 3

























