JEE Coordination Compounds PYQs
JEE Coordination Compounds PYQs are an important part of the JEE Chemistry syllabus. They help you understand the kind of questions asked from this chapter and test how well you know the main concepts, such as ligands, coordination number, oxidation state, nomenclature, isomerism, bonding, and magnetic properties.
In the exam, coordination compounds questions usually come as direct theory-based questions or simple concept-based problems. The good thing is that this chapter becomes much easier when your basics are clear. Once you understand the main ideas and rules properly, solving questions feels much more manageable. You do not need to think of coordination compounds as a very difficult chapter. With regular revision and smart practice, it can become one of the more scoring parts of JEE Chemistry.
In this blog, you will find a simple formula PDF, a section for important JEE Coordination Compounds PYQs in download format, a few practice questions with answers, and some extra questions to solve on your own. You will also learn about common mistakes students often make and a few simple tips to save time in the exam.
JEE Coordination Compounds Important PYQs PDF
This PDF can include the most important previous year questions from coordination compounds. It may cover topics like ligands, coordination number, oxidation state, IUPAC naming, Werner’s theory, types of isomerism, valence bond theory, crystal field theory, and magnetic behaviour.
Practicing these questions will help you understand the exam pattern better. It will also improve your speed, accuracy, and confidence before the exam.
Important Formulas for JEE Coordination Compounds PYQs
You only need a few important rules, ideas, and formulas to solve most coordination compounds questions in JEE. These help you understand oxidation state, naming, bonding, hybridization, magnetic nature, and stability in a much clearer way.
You can download the full formula PDF from the link above. Here is a quick look at some of the main formulas and rules:
Concept | Formula / Rule |
Oxidation State of Central Metal | Sum of oxidation states of all atoms = charge on complex |
Coordination Number | Number of ligand donor atoms attached to central metal |
Effective Atomic Number | EAN = Atomic Number of Metal − Oxidation State + Electrons Donated by Ligands |
Magnetic Moment | μ = √n(n+2) BM |
Monodentate Ligand | Ligand attached through one donor atom |
Bidentate Ligand | Ligand attached through two donor atoms |
Ambidentate Ligand | Ligand that can attach through two different atoms |
Werner’s Theory | Primary valency is ionisable, secondary valency is non-ionisable |
Naming Rule | Name ligands first, then metal with oxidation state in brackets |
Isomerism | Includes ionisation, linkage, coordination, geometrical, and optical isomerism |
These formulas and rules are often used in questions based on oxidation state, coordination number, naming of complexes, isomerism, bonding, and magnetic properties. If you revise them properly, many JEE questions start to feel much easier.
Top 5 Common Mistakes to Avoid in JEE Coordination Compounds PYQs
Many students find coordination compounds confusing because the chapter has many terms, rules, and exceptions. But in most cases, mistakes happen because small details are missed while revising or solving. Here are some common mistakes you should avoid:
Finding the wrong oxidation state
Students often forget the charges of ligands or the overall charge of the complex while finding the oxidation state of the central metal. Even one small mistake here can change the whole answer.
Mixing up coordination number and oxidation state
These two are not the same. Coordination number tells you how many donor atoms are attached to the metal, while oxidation state tells you the charge on the metal.
Making mistakes in IUPAC naming
Naming rules are very important in this chapter. Many students forget the correct order of ligands, the metal name, or the oxidation state written in brackets.
Confusing the types of isomerism
Ionisation isomerism, linkage isomerism, geometrical isomerism, and optical isomerism can seem similar at first. If the basic idea of each one is not clear, the question becomes confusing.
Ignoring magnetic behaviour and hybridization
Some students focus only on naming and oxidation state and do not pay enough attention to questions based on unpaired electrons, magnetic moment, and inner or outer orbital complexes.
List of JEE Coordination Compounds PYQs
Here is a short set of JEE-style coordination compounds questions for practice. These include common question types from oxidation state, ligands, nomenclature, isomerism, and magnetic properties. Solving them regularly can help you become faster and more confident.
Question 1
Given below are two statements:
Statement I : Crystal Field Stabilization Energy (CFSE) of $$\left[Cr\left( H_{2}O \right)_{6} \right]^{2+}$$ is greater than that of $$\left[Mn\left( H_{2}O \right)_{6} \right]^{2+}$$.
Statement II: Potassium ferricyanide has a greater spin-only magnetic moment than sodium ferrocyanide.
In the light of the above statements, choose the correct answer from the options given below :
correct answer:- 3
Question 2
Identify the metal ions among $$Co^{2+}, Ni^{2+}, Fe^{2+}, V^{3+} \text{ and } Ti^{2+}$$ having a spin-only magnetic moment value more than 3.0 BM. The sum of unpaired electrons present in the high spin octahedral complexes formed by those metal ions is __________.
correct answer:- 7
Question 3
Given below are two statements:
Statement I: Among $$[Cu(NH_{3})_{4}]^{2+},[Ni(en)_{3})]^{2+},[Ni(NH_{3})_{6}]^{2+}\text{ and }[Mn(H_{2}O)_{6}]^{2+},[Mn(H_{2}O)_{6}]^{2+}$$ has the maximum number of unpaired electrons.
Statement II : The number of pairs among $$\left\{[Ni(Cl_{4}]^{2-},[Ni(CO)_{4}]\right\},\left\{[NiCl_{4}]^{2-},[Ni(CN)_{4}]^{2-}\right\} and \left\{[Ni(CO)_{4}],[Ni(CN)_{4}]^{2-}\right\}$$ that contain only diamagnetic species is two.
ln the light of the above statements, choose the correct answer from the options given below:
correct answer:- 4
Question 4
Consider the following reactions.
$$PbCl_{2}+K_{2}CrO_{4}\rightarrow A+2KCI$$
(Hot solution)
$$A+NaOH\rightleftharpoons B+Na_{2}CrO_{4}$$
$$PbSO_{4}+4CH_{3}COONH_{4}\rightarrow (NH_{4})_{2}SO_{4}+X$$
In the above reactions, A, Band X are respectively.
correct answer:- 2
Question 5
Given below are two statements:
Statement I :
The number of species among $$SF_{4},NH_4^+,[NiCl_{4}]^{2-},XeF_{4},[PtCl_{4}]^{2-},SeF_{4}\text{ and }[Ni(CN)_{4}]^{2-}$$, tha t have tetrahedral geometry is 3.
Statement II :
In the set $$[NO_{2},BeH_{2},BF_{3},AlCl_{3}]$$ all the molecules have incomplete octet around central atom.
In the light of the above statements, choose the correct answer from the options given below :
correct answer:- 4
Question 6
$$[Ni (PPh_{3})_{2}Cl_{2} ]$$ is a paramagnetic complex. Identify the INCORRECT statements about this complex.
A. The complex exhibits geometrical isomerism.
B. The complex is white in colour.
C. The calculated spin-only magnetic moment of the complex is 2.84 BM.
D. The calculated CFSE (Crystal Field Stabilization Energy) of Ni in this complex is $$-0.8 \triangle_{0}$$
E. The geometrical arrangement of ligands in this complex is similar to that in $$Ni(CO)_{4}$$.
Choose the correct answer from the options given below :
correct answer:- 1
Question 7
The statements that are incorrect about the nickel(II) complex of dimethylglyoxime are:
A. It is red in colour.
B. It has a high solubility in water at pH =9.
C. The Ni ion has two unpaired d-electrons.
D. The N - Ni - N bond angle is almost close to 90°
E. The complex contains four five-membered metallacycles (metal containing rings).
Choose the correct answer from the options given below :
correct answer:- 4
Question 8
Given below are two statements :
Statement I: $$[CoBr_{4}]^{2-}$$ ion will absorb light of lower energy than $$[CoCl_{4}]^{2-}$$ ion.
Statement II: In $$[Col_{4}]^{2-}$$ ion, the energy separation between the two set of d-orbitals is more than $$[CoCl_{4}]^{2-}$$ ion.
ln the light of the above statements, choose the correct answer from the options given below:
correct answer:- 3
Question 9
The crystal field splitting energy of $$[Co(oxalate)_{3}]^{3-}$$ complex is 'n' times that of the $$[Cr(oxalate)_{3}]^{3-}$$ complex. Here 'n' is_______. (Assume d $$\triangle_{\circ} > > P$$)
correct answer:- 2
Question 10
A first row transition metal (M) does not liberate $$H_{2}$$ gas from dilute HCI. 1 mol of aqueous solution of $$MSO_{4}$$ is treated with excess of aqueous KCN and then $$H_{2} S(g)$$ is passed through the solution. The amount of MS (metal sulphide) formed from the above reaction is _______ mol.
correct answer:- 3
Question 11
Two p-block elements $$X$$ and $$Y$$ form fluorides of the type $$EF_{3}$$. The fluoride compound $$XF_{3}$$ is a Lewis acid and $$YF_{3}$$ is a Lewis base. The hybridizations of the central atoms of $$XF_{3}$$ and $$YF_{3}$$ respectively are
correct answer:- 2
Question 12
Given below are h¥o statements:
Statement I: Hybridisation, shape and spin only magnetic moment of $$K_{3}[Co(CO_{3})_{3}]$$ is $$sp^{3}d^{2}$$, octahedral and 4.9 BM respectively.
Statement II: Geometty, hybridisation and spin only magnetic moment values (BM) of the ions $$[Ni(CN)_{4}]^{2-},[MnBr_{4}]^{2-}\text{ and }[CoF_{6}]^{3-}$$ respectively are square planar, tetrahedral, octahedral;$$dsp^{2},sp^{3},sp^{3}d^{2}$$ and 0, 5.9, 4.9.
In the light of the above statements, choose the correct answer from the options given below
correct answer:- 2
Question 13
Consider a mixtm-e 'X' which is made by dissolving 0.4 mol of $$[Co(NH_{3})_{5}SO_{4}]Br$$ and 0.4 mol of $$[Co(NH_{3})_{5}Br]SO_{4}$$ in water to make 4 L of solution. When 2 L of mixture 'X' is allowed to react with excess of $$AgNO_{3}$$,it fonns precipitate 'Y'. The rest 2 L of mixture 'X' reacts with excess $$BaCl_{2}$$ to fonn precipitate 'Z'. Which of the following statements is CORRECT?
correct answer:- 2
Question 14
Given below are two statements:
Statement I: The number of paramagnetic species among $$[CoF_{6}]^{3-},[TiF_{6}]^{3-},V_{2}O_{5}\text{ and }[Fe(CN)_{6}]^{3-}$$ is 3.
Statement II: $$K_{4}[Fe(CN)_{6}] < K_{3}[Fe(CN)_{6}] < [Fe(H_{2}O)_{6}]SO_{4}.H_{2}O < [Fe(H_{2}O)_{6}]Cl_{3}$$ is the correct order in terms of number of unpaired electron(s) present in the complexes.
In the light of the above statements, choose the correct answer from the options given below
correct answer:- 1
Question 15
Among the following, the CORRECT combinations are
$$\text{A. IF}_{3} \rightarrow \text{T-shaped}(\text{sp}^{3}\text{d})$$
$$\text{B. IF}_{5} \rightarrow \text{Square pyramidal(sp}^{3}\text{d}^{2})$$
$$\text{C. IF}_{7} \rightarrow \text{Pentagonal bipyrnmidal(sp}_{3}\text{d}^{3})$$
$$\text{D. ClO}_{4}^{-} \rightarrow \text{Square planar(sp}^{3}\text{d})$$
Choose the correct answer from the options given below:
correct answer:- 2
Question 16
The wavelength of light absorbed for the following complexes are in the order
(I)$$\left[Co{(NH_{3})}_{6}\right]^{3+}$$
(II)$$\left[Co{(H_{2}O)}_{6}\right]^{3+}$$
(III)$$\left[Co{(CN)}_{6}\right]^{3-}$$
(IV)$$\left[Co(NH_{3})_{5}(H_{2}O)\right]^{3+}$$
(V)$$\left[CoF_{6}\right]^{3-}$$
correct answer:- 3
Question 17
A chromium complex with a formula $$CrCl_{3}.6H_{2}O$$ has a spin only magnetic moment value of 3.87 BM and its solution conductivity corresponds to 1:2 electrolyte. 2.75 g of the complex solution was initially passed through a cation exchanger. The solution obtained after the process was reacted with excess of $$AgNO_{3}$$. The amount of AgCI formed in the above process is __ g. (Nearest
integer)
[Given: Molar massing $$mol^{-1}$$ Cr:52; Cl:35.5, Ag:108, 0:16, H:1]
correct answer:- 3
Question 18
The correct statement among the followring is:
correct answer:- 2
Question 19
X is the number of geometrical isomers exhibited by $$[Pt(NH_{3})(H_{2}O)BrCl]$$.
Y is the number of optically inactive isomer(s) exhibited by $$[CrCl_{2}(ox)_{2}]^{3-}$$
z is the number of geometrical isomers exhibited by $$[Co(NH_{3})_{3}(NO_{2})_{3}]$$.
The value of X + Y + Z is________.
correct answer:- 6
Question 20
Identify the CORRECT set of details from the following:
A. $$[Co(NH_3)_6]^{3+}$$ : Inner orbital compex; $$d^{2}sp^{3}$$ hybridized
B. $$[MnCl_6]^{3-}$$ : Outer orbital complex; $$sp^{3}d^{2}$$ hybridized
C. $$[CoF_6]^{3-}$$ : Outer orbital complex; $$d^{2}sp^{3}$$ hybridized
D. $$[FeF_6]^{3-}$$ : Outer orbital complex; $$sp^{3}d^{2}$$ hybridized
E. $$[Ni(CN)_4]^{2-}$$ : Inner orbital complex; $$sp^{3}d^{2}$$ hybridized
Choose the correct answer from the options given below:
correct answer:- 4
Question 21
Total number of unpaired electrons present in the central met al atoms/ions of
$$[Ni(CO)_4],[NiCl_4]^{2-},[PtCl_2(NH_3)_2],[Ni(CN)_4]^{2-} \text {and } [Pt(CN)_4]^{2-}$$ is_____
correct answer:- 2
Question 22
The correct increasing o rd e r of spin-only magnetic moment values of the complex ions $$[MnBr_{4}]^{2-}(A),[Cu(H_{2}O)_{6}]^{2+}(B),[Ni(CN)_{4}]^{2-}(C)\text{ and }[Ni(H_{2}O)_{6}]^{2+}(D)$$ is:
correct answer:- 4
Question 23
Match List - I with List - Il according to shape.
List - I List - II
A. $$ XeO_3 $$ I. $$ BrF_5 $$
B. $$ XeF_2 $$ II. $$ NH_3 $$
C. $$ XeO_2 F_2 $$ III. $$ [I_3]^-{} $$
D. $$ XeOF_4 $$ IV. $$ SF_4 $$
Choose the correct answer from the options given below:
correct answer:- 2
Question 24
Consider the following statements about manganate and permanganate ions. Identify the correct statements.
A. The geometry of both manganate and permanganate ions is tetrahedral
B. The oxidation states of Mn in manganate and permanganate are + 7 and + 6, respectively.
C. Oxidation of Mn(II) salt by peroxodisulphate gives manganate ion as the final product.
D. Manganate ion is paramagnetic and permanganate ions is diamagnetic.
E. Acidified permanganate ion reduces oxalate, nitrite and iodide ions.
Choose the correct answer from the options given below :
correct answer:- 1
Question 25
The numbei of isoelectronic species among $$Sc^{3+},Cr^{2+},Mn^{3+},Co^{3+}\text{ and }Fe^{3+}$$ is 'n'. If 'n' moles of AgCl is formed during the reaction of complex with formula $$CoCl_{3}(en)_{2}NH_{3}$$ with excess of $$AgNO_{3}$$ solution, then the number of electrons present in the $$t_{2g}$$ orbital of the complex is ________.
correct answer:- 6
Question 26
The correct order of the following complexes in terms of their crystal field stabilization energies is :
correct answer:- 4
Question 27
Identify the homoleptic complex(es) that is/are low spin. $$(A)[Fe(CN)_{5}NO]^{2-}(B)[CoF_{6}]^{3-}(C)[Fe(CN)_{6}]^{4-}(D)[Co(NH_{3})_{6}]^{3+}(E)[Cr(H_{2}O)_{6}]^{2+}$$ Choose the correct answer from the options given below :
correct answer:- 2
Question 28
The complex of $$Ni^{2+}$$ ion and dimethyl glyoxime contains ________ number of Hydrogen (H) atoms.
correct answer:- 14
Question 29
In which of the following complexes the CFSE, $$\triangle_o$$ will be equal to zero?
correct answer:- 4
Question 30
From the magnetic behaviour of $$[NiCl_4]^{2-} \text{(paramagnetic) and } [Ni(CO)_4] $$ (diamagnetic), choose the correct geometry and oxidation state.
correct answer:- 3
Question 31
Identify the coordination complexes in which the central metal ion has $$d^{4}$$ configuration.
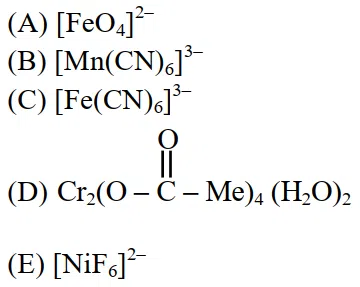
Choose the correct answer from the options given below :
correct answer:- 3
Question 32
One mole of the octahedral complex compound $$Co(NH_3)_5Cl_3$$ gives 3 moles of ions on dissolution in water. One mole of the same complex reacts with excess $$AgNO_3$$ solution to yield two moles of $$AgCl(s).$$ The structure of the complex is:
correct answer:- 3
Question 33
Find the compound ' A ' from the following reaction sequences.
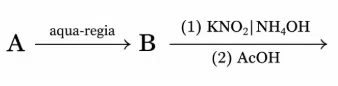
yellow ppt
correct answer:- 1
Question 34
{The conditions and consequence that favours the $$t_{2gg} e_g^1$$ configuration in a metal complex are:
correct answer:- 2
Question 35
The metal ion whose electronic configuration is not affected by the nature of the ligand and which gives a violet colour in non-luminous flame under hot condition in borax bead test is
correct answer:- 3
Question 36

Choose the correct answer from the options given below :
correct answer:- 1
Question 37
The correct increasing order of stability of the complexes based on $$\Delta_{\circ}$$ value is: I.$$[Mn(CN)_{6}]^{3}$$ II.$$[Co(CN)_{6}]^{4-}$$ III.$$[Fe(CN)_{6}]^{4-}$$ IV.$$[Fe(CN)_{6}]^{3-}$$
correct answer:- 2
Question 38
The complex that shows Facial - Meridional isomerism is:
correct answer:- 3
Question 39
$$CrCl_{3}\cdot xNH_{3}$$ can exist as a complex. 0.1 molal aqueous solution of this complex shows a depression in freezing point of $$0.558^{\circ}C$$. Assuming 100% ionisation of this complex and coordination number of Cr is 6 , the complex will be (Given $$K_{f}$$ = 1.86 K kg $$mol^{-1}$$)
correct answer:- 1
Question 40
The d- electronic configuration of an octahedral Co(II) complex having magnetic moment of 3.95 BM is:
correct answer:- 3
Question 41
Identify the homoleptic complexes with odd number of d electrons in the central metal : (A) $$[FeO_4]^{2-}$$ (B) $$[Fe(CN)_6]^{3-}$$ (C) $$ [Fe(CN)_5NO]^{2-}$$ (D) $$[CoCl_4]^{2-}$$ (E) $$ [Co(H_2O)_3F_3]$$ Choose the correct answer from the options given below :
correct answer:- 3
Question 42
The calculated spin-only magnetic moments of $$K_3[Fe(OH)_6]$$ and $$K_4[Fe(OH)_6]$$ respectively are :
correct answer:- 4
Question 43
Consider the following low-spin complexes $$K_3[Co(NO_2)_6],\; K_4[Fe(CN)_6],\; K_3[Fe(CN)_6],\; Cu_2[Fe(CN)_6] and Zn_2[Fe(CN)_6]$$ The sum of the spin-only magnetic moment values of
complexes having yellow colour is. _____ B.M. (answer in nearest integer)
correct answer:- 0
Question 44
Number of complexes from the following with even number of unpaired "d" electrons is: $$[V(H_2O)_6]^{3+},\ [Cr(H_2O)_6]^{2+},\ [Fe(H_2O)_6]^{3+},\ [Ni(H_2O)_6]^{3+},\ [Cu(H_2O)_6]^{2+}$$. [Given atomic numbers: V=23, Cr=24, Fe=26, Ni=28, Cu=29]
correct answer:- 1
Question 45
The correct sequence of ligands in the order of decreasing field strength is:
correct answer:- 2
Question 46
Match List - I with List - II.

Choose the correct answer from the options given below :
correct answer:- 4
Question 47
Which of the following complex is homoleptic?
correct answer:- 1
Question 48
Given below are two statements: Statement (I): A solution of $$[Ni(H_2O)_6]^{2+}$$ is green in colour. Statement (II): A solution of $$[Ni(CN)_4]^{2-}$$ is colourless. In the light of the above statements, choose the most appropriate answer from the options given below:
correct answer:- 2
Question 49
Given below are two statements:
Statement (I): Dimethyl glyoxime forms a six membered covalent chelate when treated with $$NiCl_2$$ solution in presence of $$NH_4OH$$.
Statement (II): Prussian blue precipitate contains iron both in +2 and +3 oxidation states.
In the light of the above statements, choose the most appropriate answer from the options given below:
correct answer:- 1
Question 50
$$[Co(NH_3)_6]^{3+}$$ and $$[CoF_6]^{3-}$$ are respectively known as:
correct answer:- 2

 Group
Group






.webp)















