JEE Equilibrium PYQs
JEE Equilibrium PYQs are an important part of the JEE Chemistry syllabus. They help you understand the kind of questions asked from this chapter and check how well you know the main ideas, such as chemical equilibrium, ionic equilibrium, the law of mass action, equilibrium constant, Le Chatelier’s principle, pH, buffer solutions, and solubility product.
In the exam, equilibrium questions usually come as direct numerical problems or simple concept-based questions. The good thing is that this chapter becomes much easier when your basics are clear. Once you understand the concepts properly and know which formula or rule to use, solving questions feels much more manageable. You do not need to think of equilibrium as a very difficult chapter. With regular revision and smart practice, it can become one of the more scoring parts of JEE Chemistry.
In this blog, you will find a simple formula PDF, a section for important JEE Equilibrium PYQs in download format, a few practice questions with answers, and some extra questions to solve on your own. You will also learn about common mistakes students often make and a few simple tips to save time in the exam.
JEE Equilibrium Important PYQs PDF
This PDF can include the most important previous year questions from equilibrium. It may cover topics like dynamic equilibrium, equilibrium constant, reaction quotient, Le Chatelier’s principle, acids and bases, ionic equilibrium, pH, pOH, hydrolysis, buffer solution, and solubility product.
Practicing these questions will help you understand the exam pattern better. It will also improve your speed, accuracy, and confidence before the exam.
Important Formulas for JEE Equilibrium PYQs
You only need a few important formulas and ideas to solve most equilibrium questions in JEE. These formulas help you understand concentration changes, pH values, equilibrium constants, and ionic calculations more clearly.
You can download the full formula PDF from the link above. Here is a quick look at some of the main formulas:
Concept | Formula |
Equilibrium Constant | Kc = [Products] / [Reactants] |
Equilibrium Constant in Pressure Terms | Kp = [Partial Pressure of Products] / [Partial Pressure of Reactants] |
Relation Between Kp and Kc | Kp = Kc(RT)ⁿ |
pH | pH = -log[H⁺] |
pOH | pOH = -log[OH⁻] |
Ionic Product of Water | Kw = [H⁺][OH⁻] |
Relation Between pH and pOH | pH + pOH = 14 |
Henderson Equation for Buffer | pH = pKa + log([Salt]/[Acid]) |
Solubility Product | Ksp = [Ion₁]ᵃ [Ion₂]ᵇ |
Degree of Dissociation | α = Number Dissociated / Total Number |
Ostwald’s Dilution Law | Ka = Cα² / (1 - α) |
These formulas are commonly used in questions based on chemical equilibrium, ionic equilibrium, acids and bases, buffer solutions, and solubility product. If you revise them properly, many JEE questions will start to feel much easier.
Top 5 Common Mistakes to Avoid in JEE Equilibrium PYQs
Many students find equilibrium confusing at first because it includes both concepts and numerical formulas. But most mistakes happen because small details are missed while solving. Here are some common mistakes you should avoid:
Mixing up Kc and Kp
Kc is written in terms of concentration, while Kp is written in terms of partial pressure. Many students confuse the two and end up using the wrong formula.
Forgetting the difference between strong and weak acids or bases
Strong acids and bases ionize almost completely, while weak ones do not. If this basic idea is not clear, pH questions become confusing.
Making mistakes in pH and pOH calculations
Students often forget the log relation or the formula pH + pOH = 14. Even a small calculation mistake can change the whole answer.
Not applying Le Chatelier’s principle correctly
In equilibrium questions, changes in concentration, pressure, or temperature can shift the equilibrium. Many students know the rule but do not apply it properly in actual questions.
Confusing solubility with solubility product
Solubility tells how much of a substance dissolves, while Ksp is the equilibrium constant for its dissolution. They are related, but they are not the same thing.
List of JEE Equilibrium PYQs
Here is a short set of JEE-style equilibrium questions for practice. These include common question types from equilibrium constant, pH, ionic equilibrium, and solubility product. Solving them regularly can help you become faster and more confident.
Question 1
The first and second ionization constants of $$H_{2}X$$ are $$2.5 \times 10^{-8}$$ and $$1.0 \times 10^{-13}$$ respectively. The concentration of $$X^{2-}$$ in $$0.1 MH_{2} X$$ solution is _________ $$\times 10^{-15}M$$. (Nearest Integer)
correct answer:- 100
Question 2
Which of the followmg mixture gives a buffer solution with pH=9.25?
Given : $$pK_{b}$$ $$(NH_{4}OH)$$ = 4.75
correct answer:- 4
Question 3
For the following gas phase equilibrium reaction at constant temperature,
$$NH_{3}(g)\rightleftharpoons 1/2N_{2}(g)+3/2H_{2}(g)$$
if the to tal pressure is $$\sqrt{3}$$ atm and the pressure equilibrium constant ($$K_{p}$$) is 9 atm, then the degree of dissociation is given as $$(x\times 10^{-2})^{-1/2}$$.The value of x is ______. (nearest integer)
correct answer:- 125
Question 4
Dissociation of a gas $$A_{2}$$ takes place according to the following chemical reaction.
At equilibrium, the total pressure is 1 bar at 300K.
$$A_{2}(g)\rightleftharpoons 2A(g)$$
The standard Gibbs energy of formation of the involved substances has been
provided below:
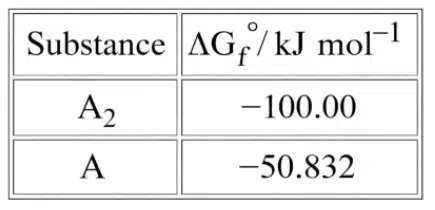
The degree of dissociation of $$A_{2} (g)$$ is given by $$(x\times10^{-2})^{1/2} $$ where $$x$$ =
_____ . (Nearest integer).
[Given: $$R=8 J \text{ }mol^{-1}K^{-1},\log{2}=0.3010, \log {3}=0.48]$$
Assume degree of dissociation is not negligible.
correct answer:- 33
Question 5
Consider two Group IV metal ious $$X^{2+}\text{ and }Y^{2+}$$
A solution containing $$0.01 M X^{2+}\text{ and }0.01MY^{2+}$$ is satmated with $$H_{2}S$$. The pH at which the metal sulphide YS will form as a precipitate is __ . (Nearest integer)
$$(Given:K_{sp}(XS)=1\times 10^{-22} \text{ at } 25^{\circ}C,K_{sp}(YS)=4\times 10^{-16} \text{ at } 25^{\circ}C,[H_{2}S]=0.1M\text{ in solution },K_{a1}\times K_{a2}(H_{2}S)=1.0\times 10^{-21},\log{2}=0.30,\log{3}=0.48,\log{5}=0.70)$$
correct answer:- 4
Question 6
Consider the following gaseous equilibrimn in a closed container of volume 'V' at T(K).
$$P_{2}(g)+Q_{2}(g)\rightleftharpoons 2PQ(g)$$
2 moles each of $$P_{2}(g)$$, $$Q_{2}(g)$$ and PQ(g) are present at equilibrium. Now one mole each of'$$P_{2}$$' and '$$Q_{2}$$' are added to the equilibrium keeping the temperature at T(K). The number of moles of $$P_{2}$$, $$Q_{2}$$ and PQ at the new equilibrium, respectively, are
correct answer:- 1
Question 7
Molar conductivity of a weak acid HQ of concentration 0.18 M was found to be 1/30 of the molar conductivity of another weak acid HZ with concentration of 0.02M. If $$\lambda^{\circ}{}_{Q}-$$ happened to be equal with $$\lambda^{\circ}{}_{Z}-$$, then the difference of the $$pK_{a}$$ values of the two weak acids $$(pK_{a}(HQ) - pK_{a}(HZ))$$ is ___ (Nearest integer).
[Given: degree of dissociation ($$\alpha$$) << 1 for both weak acids, $$\lambda^{\circ}$$ : limiting molar conductivity of ions]
correct answer:- 2
Question 8
Consider a weak base 'B' of $$pK_{b}=5.699 $$. 'x' mL of 0.02 M HCI and 'y' mL of 0.02 M weak base 'B' are mixed to make 100 mL of a buffer of pH 9 at 25 °C. The values of 'x' and 'y' respectively are:
[Given: log 2 = 0.3010, log 3 = 0.4771, log 5 = 0.699]
correct answer:- 2
Question 9
Consider the dissociation equilibrium of the following weak acid $$HA\rightleftharpoons H^{+}(aq)+A^{-}(aq)$$If the pKa of the acid is 4, then the pH of 10 mM HA solution is __ .(Nearest integer)
[Given: The degree of dissociation can be neglected with respect to unity]
correct answer:- 3
Question 10
$$X_2(g ) + Y_2(g ) \rightleftharpoons 2Z(g)$$
$$X_2(g )$$ and $$Y_2(g )$$ are added to a 1 L flask and it is found that the system attains the above equilibrium at T(K) with the number of moles of $$X_2(g ),\text{ } Y_2(g )$$ and $$Z(g)$$ being 3, 3 and 9 mol respectively (equilibrium moles). Under this condition of equilibrium, 10 mol of $$Z(g$$) is added to the flask and the temperature is maintained at $$T(K)$$. Then the number of moles of $$Z(g)$$ in the flask when the new equilibrium is established is __ . (Nearest integer)
correct answer:- 15
Question 11
Observe the following equilibrium in a 1 L flask.
$$A(g)\rightleftharpoons B(g)$$
At T(K), the equilibrium concentrations of A and B are 0.5 Mand 0.375 M respectively. 0.1 moles of A is added into the flask and heated to T(K) to establish the equilibrium again. The new equilibrium concentrations (in M) of A and B are respectively
correct answer:- 1
Question 12
The molar solubility(s) of zirconium phosphate with molecular formula $$(Zr^{4+})_{3}(PO_{4}^{3-})_{4}$$ is given by relation :
correct answer:- 2
Question 13
A vessel at 1000 K contains $$CO_2$$ with a pressure of 0.5 atm . Some of $$CO_2$$ is converted into CO on addition of graphite. If total pressure at equilibrium is 0.8 atm , then Kp is :
correct answer:- 1
Question 14
Consider the reaction $$X_{2}Y(g)=X_{2}(g)+\frac{1}{2}Y_{2}(g)$$ The equation representing correct relationship between the degree of dissociation (x) of $$X_{2}Y(g)$$ with its equilibrium constant Kp is ______ . Assume x to be very very small.
correct answer:- 2
Question 15
pH of water is 7 at $$25^{\circ}C$$.If water is heated to $$80^{\circ}C$$.,it's pH will :
correct answer:- 1
Question 16
$$ 37.8\,g\,N_2O_5$$ was taken in a $$1\,L$$ reaction vessel and allowed to undergo the following reaction at 500 K $$2N_2O_5(g) \rightleftharpoons 2N_2O_4(g) + O_2(g)$$ The total pressure at equilibrium was found to be $$18.65\,$$bar. Then, $$K_p = \underline{\hspace{2cm}}\times10^{-2}$$ [nearest integer]. Assume $$N_2O_5$$ to behave ideally under these conditions. Given: $$R=0.082\,bar\,L\,mol^{-1}K^{-1}$$
correct answer:- 962
Question 17
$$\text{For the reaction, } H_2(g) + I_2(g) \rightleftharpoons 2HI(g),$$ Attainment of equilibrium is predicted correctly by:
correct answer:- 2
Question 18
A weak acid HA has degree of dissociation x . Which option gives the correct expression of $$(pH pK_{a})$$ ?
correct answer:- 4
Question 19
At temperature $$T$$, compound $$AB_{2(g)}$$ dissociates as $$AB_{2} \rightleftharpoons AB_{(g)}+\frac{1}{2}B_{2(g)}$$ having degree of dissociation $$x$$ (small compared to unity). The correct expression for $$x$$ in terms of $$K_{p}$$ and p is
correct answer:- 3
Question 20
Which of the following happens when $$NH_{4}OH$$ is added gradually to the solution containing 1 M $$A^{2+}$$ and $$1MB^{3+}$$ ions? Given : $$K_{sp}[A(OH)_{2}]= 9 \times 10^{-10}$$ and $$K_{sp}[B(OH)_{3}]= 27 \times 10^{-18}$$ at 298 K .
correct answer:- 3
Question 21
If 1 mM solution of ethylamine produces pH=9, then the ionization constant $$(K_{b})$$ of ethylamine is $$10^{-x}$$. The value of is ______ (nearest integer). [The degree of ionization of ethylamine can be neglected with respect to unity.]
correct answer:- 7
Question 22
Consider the equilibrium $$CO(g)+3H_{2}(g)\rightleftharpoons CH_{4}(g)+H_{2}O(g)$$ If the pressure applied over the system
increases by two fold at constant temperature then (A) Concentration of reactants and products increases. (B) Equilibrium will shift in forward direction. (C) Equilibrium constant increases since concentration of products increases. (D) Equilibrium constant remains unchanged as concentration of reactants and products remain same. Choose the correct answer from the options given below :
correct answer:- 2
Question 23
What will be the decreasing order of basic strength of the following conjugate bases? $$^-OH,\ R\bar{O},\ CH_3CO\bar{O},\ C\bar{l}$$
correct answer:- 1
Question 24
In the precipitation of the iron group (III) in qualitative analysis, ammonium chloride is added before adding ammonium hydroxide to:
correct answer:- 4
Question 25
Arrange the following in increasing order of solubility product :$$\mathrm{Ca(OH)_2, \ AgBr, \ PbS, \ HgS}$$
correct answer:- 4
Question 26
$$K_a$$ for $$CH_3COOH$$ is $$1.8 \times 10^{-5}$$ and $$K_b$$ for $$NH_4OH$$ is $$1.8 \times 10^{-5}$$. The pH of ammonium acetate solution will be:
correct answer:- 7
Question 27
Solubility of calcium phosphate (molecular mass, M) in water is W g per 100 mL at 25°C. Its solubility product at 25°C will be approximately.
correct answer:- 2
Question 28
Which of the following is strongest Bronsted base?
correct answer:- 4
Question 29
Given below are two statements : Statement (I) : Aqueous solution of ammonium carbonate is basic.
Statement (II) : Acidic/basic nature of salt solution of a salt of weak acid and weak base depends on $$K_a$$ and $$K_b$$ value of acid and the base forming it.
In the light of the above statements, choose the most appropriate answer from the options given below :
correct answer:- 1
Question 30
For the reaction $$N_2O_4(g) \rightleftharpoons 2NO_2(g)$$, $$K_p = 0.492$$ atm at $$300$$ K. $$K_c$$ for the reaction at same temperature is _______ $$\times 10^{-2}$$. (Given: $$R = 0.082$$ L atm mol$$^{-1}$$ K$$^{-1}$$)
correct answer:- 2
Question 31
The following concentrations were observed at $$500$$ K for the formation of $$NH_3$$ from $$N_2$$ and $$H_2$$. At equilibrium: $$[N_2] = 2 \times 10^{-2}$$ M, $$[H_2] = 3 \times 10^{-2}$$ M and $$[NH_3] = 1.5 \times 10^{-2}$$ M. Equilibrium constant for the reaction is ______.
correct answer:- 417
Question 32
The pH at which $$Mg(OH)_2 [K_{sp} = 1 \times 10^{-11}]$$ begins to precipitate from a solution containing $$0.10 \text{ M } Mg^{2+}$$ ions is ______.
correct answer:- 9
Question 33
The pH of an aqueous solution containing 1M benzoic acid ($$pK_a = 4.20$$) and 1M sodium benzoate is 4.5. The volume of benzoic acid solution in 300 mL of this buffer solution is _________ mL.
correct answer:- 100
Question 34
For the given reaction, choose the correct expression of $$K_C$$ from the following: $$\text{Fe}^{3+}_{(aq)} + \text{SCN}^-_{(aq)} \rightleftharpoons (\text{FeSCN})^{2+}_{(aq)}$$
correct answer:- 1
Question 35
$$A_g \rightleftharpoons B_g + \frac{C}{2}_g$$. The correct relationship between $$K_P$$, $$\alpha$$ and equilibrium pressure $$P$$ is
correct answer:- 2
Question 36
The equilibrium constant for the reaction $$SO_3(g) \rightleftharpoons SO_2(g) + \frac{1}{2}O_2(g)$$ is $$K_c = 4.9 \times 10^{-2}$$. The value of $$K_c$$ for the reaction given below is $$2SO_2(g) + O_2(g) \rightleftharpoons 2SO_3(g)$$:
correct answer:- 4
Question 37
Given below are two statements : Statement I : On passing $$HCl_{(g)}$$ through a saturated solution of $$BaCl_2$$, at room temperature white turbidity appears. Statement II : When HCl gas is passed through a saturated solution of NaCl, sodium chloride is precipitated due to common ion effect. In the light of the above statements, choose the most appropriate answer from the options given below :
correct answer:- 2
Question 38
Consider the given chemical reaction sequence :
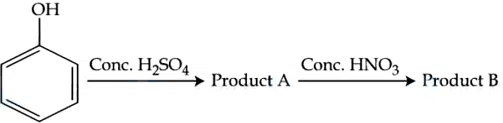
Total sum of oxygen atoms in Product A and Product B are ______
correct answer:- 14
Question 39
Consider the given reaction, identify the major product "P". $$CH_3 - COOH \xrightarrow{(i) LiAlH_4 \; (ii) PCC \; (iii) HCN/OH^{-} \; (iv) H_2O/OH^{-}, \Delta}$$ "P"
correct answer:- 3
Question 40
The correct sequence of acidic strength of the following aliphatic acids in their decreasing order is: $$CH_3CH_2COOH, CH_3COOH, CH_3CH_2CH_2COOH, HCOOH$$
correct answer:- 3

 Group
Group






.webp)












