Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Dissociation of a gas $$A_{2}$$ takes place according to the following chemical reaction.
At equilibrium, the total pressure is 1 bar at 300K.
$$A_{2}(g)\rightleftharpoons 2A(g)$$
The standard Gibbs energy of formation of the involved substances has been
provided below:

The degree of dissociation of $$A_{2} (g)$$ is given by $$(x\times10^{-2})^{1/2} $$ where $$x$$ =
_____ . (Nearest integer).
[Given: $$R=8 J \text{ }mol^{-1}K^{-1},\log{2}=0.3010, \log {3}=0.48]$$
Assume degree of dissociation is not negligible.
Correct Answer: 33
$$K_p = \frac{4\alpha^2}{1-\alpha^2} \times P$$
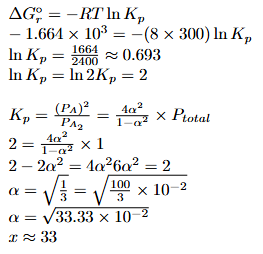
Create a FREE account and get:
Educational materials for JEE preparation
