Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
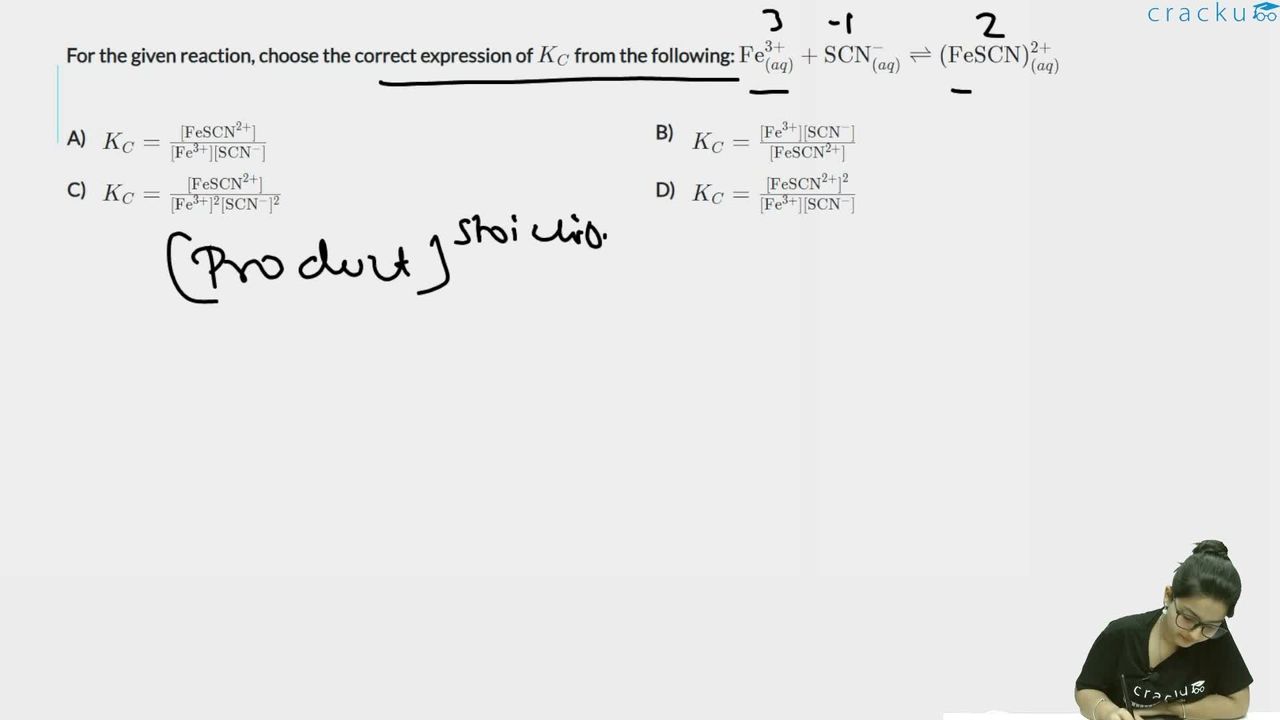
For the given reaction, choose the correct expression of $$K_C$$ from the following: $$\text{Fe}^{3+}_{(aq)} + \text{SCN}^-_{(aq)} \rightleftharpoons (\text{FeSCN})^{2+}_{(aq)}$$
We need to write the equilibrium constant expression ($$K_C$$) for the reaction:
$$ Fe^{3+}_{(aq)} + SCN^-_{(aq)} \rightleftharpoons (FeSCN)^{2+}_{(aq)} $$
For a general reaction $$aA + bB \rightleftharpoons cC + dD$$ in solution, the equilibrium constant in terms of molar concentrations is:
$$ K_C = \frac{[C]^c[D]^d}{[A]^a[B]^b} $$
Products go in the numerator and reactants go in the denominator, each raised to the power of their stoichiometric coefficient.
Here, the stoichiometric coefficients are all 1:
- Product: $$[FeSCN^{2+}]$$ with coefficient 1
- Reactants: $$[Fe^{3+}]$$ with coefficient 1 and $$[SCN^-]$$ with coefficient 1
$$ K_C = \frac{[FeSCN^{2+}]}{[Fe^{3+}][SCN^-]} $$
The correct answer is Option (1): $$K_C = \frac{[FeSCN^{2+}]}{[Fe^{3+}][SCN^-]}$$.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
