JEE Atomic Structure PYQs
JEE Atomic Structure PYQs are an important part of the JEE Chemistry syllabus. They help you understand the type of questions asked from this chapter and how well you know the main concepts, such as atomic models, subatomic particles, electronic configuration, quantum numbers, Bohr’s theory, spectra, and de Broglie’s principle.
In the exam, atomic structure questions usually appear as direct theory-based questions or simple numericals. The good thing is that this chapter becomes much easier when your basics are clear. Once you understand the main ideas and formulas properly, solving questions feels much more manageable. You do not need to think of atomic structure as a difficult chapter. With regular revision and smart practice, it can become one of the more scoring parts of JEE Chemistry.
In this blog, you will find a simple formula PDF, a section for important JEE Atomic Structure PYQs in download format, a few practice questions with answers, and some extra questions to solve on your own. You will also learn about common mistakes students often make and a few simple tips to save time in the exam.
JEE Atomic Structure Important PYQs PDF
This PDF can include the most important previous year questions from atomic structure. It may cover topics like Thomson’s model, Rutherford’s model, Bohr’s atomic model, electronic configuration, quantum numbers, wave nature of electrons, Heisenberg’s uncertainty principle, and hydrogen spectrum.
Practicing these questions will help you understand the exam pattern better. It will also improve your speed, accuracy, and confidence before the exam.
Important Formulas for JEE Atomic Structure PYQs
You only need a few important formulas, rules, and ideas to solve most atomic structure questions in JEE. These help you understand energy levels, electron movement, wavelength, frequency, and quantum numbers more clearly.
You can download the full formula PDF from the link above. Here is a quick look at some of the main formulas and rules:
Concept | Formula / Rule |
Energy of Electron in Bohr Orbit | Eₙ = -13.6 / n² eV |
Radius of nth Orbit | rₙ = 0.529 × n² / Z Å |
Velocity of Electron | vₙ = 2.18 × 10⁶ × Z / n m/s |
de Broglie Wavelength | λ = h / mv |
Energy of Photon | E = hν |
Relation Between Speed and Frequency | c = νλ |
Maximum Electrons in Shell | 2n² |
Maximum Electrons in Subshell | 4l + 2 |
Uncertainty Principle | Δx · Δp ≥ h / 4π |
Number of Orbitals in a Shell | n² |
These formulas and rules are often used in questions based on Bohr’s model, spectra, quantum numbers, electronic arrangement, and the wave nature of electrons. If you revise them properly, many JEE questions will feel much easier to solve.
Top 5 Common Mistakes to Avoid in JEE Atomic Structure PYQs
Many students find atomic structure confusing because it includes both theory and formulas. But most mistakes happen because small details are missed while revising or solving. Here are some common mistakes you should avoid:
Mixing up orbit and orbital
Orbit and orbital are not the same. An orbit comes from Bohr’s model, while an orbital is a region where the chance of finding an electron is high. Many students confuse these two terms.
Forgetting the meaning of quantum numbers
Each quantum number gives different information about an electron. If you mix up principal, azimuthal, magnetic, and spin quantum numbers, the answer can easily go wrong.
Using the wrong formula in Bohr model questions
Questions based on radius, energy, or velocity need the correct Bohr formula. Many students remember the chapter but apply the wrong formula in numerical problems.
Ignoring units in wavelength and energy questions
Atomic structure often includes values in angstrom, nanometre, electron volt, and joule. If you do not convert units carefully, the final answer may become incorrect.
Not revising electronic configuration rules properly
Aufbau principle, Pauli exclusion principle, and Hund’s rule are basic but very important. If these are not clear, many direct questions become confusing.
List of JEE Atomic Structure PYQs
Here is a short set of JEE-style atomic structure questions for practice. These include common question types from Bohr’s model, quantum numbers, electronic configuration, and de Broglie principle. Solving them regularly can help you become faster and more confident.
Question 1
Consider the following spectral lines for atomic hydrogen :
A. First line of Paschen series
B. Second line of Balmer series
C. Third line of Paschen series
D. Fourth line of Bracket series
The correct arrangement of the above lines in ascending order of energy is :
correct answer:- 2
Question 2
Given below are two statements:
Statement I : When an electric discharge is passed through gaseous hydrogen, the hydrogen molecules dissociate and the energetically excited hydrogen atoms produce electromagnetic radiation of discrete frequencies.
Statement II: The frequency of second line of Balmer series obtained from He+ is equal to that of first line of Lyman series obtained from hydrogen a tom.
ln the light of the above statements, choose the correct answer from the options given below :
correct answer:- 2
Question 3
The energy of first (lowest) Balmer line of H atom is x J. The energy (in J) of second Balmer line of H atom is :
correct answer:- 2
Question 4
Which of the following statements regarding the energy of the stationary state is true in the following one - electron systems?
correct answer:- 1
Question 5
Given,
(A) $$n = 5, m_{1} = -1$$
(B) $$n = 3, 1 = 2, m_{1} = -1, m_{2} = +\frac{1}{2}$$
The maximum number of electron(s) in an atom that can have the quantum numbers as given in (A) and (B) respectively are:
correct answer:- 2
Question 6
The energy required by electrons, present in the first Bohr orbit of hydrogen atom to J $$ mol^{-1}C $$ be excited to second Bohr orbit is ______ .
Given $$ R_{H}=2.18\times 10^{-11} $$
correct answer:- 3
Question 7
The hydrogen spectnun consists of several spectral lines in Lyman series ($$L_{1},L_{2},L_{3}....;L_{1}$$ has lowest energy among Lyman series). Similarly it consists of several spectral lines in Balmer series($$B_{1},B_{2},B_{3}....;B_{1}$$ has lowest energy among Balmer lines). The energy of $$L_{1}$$ is x times the energy of $$B_{1}$$. The value of x is_____ $$\times 10^{-1}$$. (Nearest integer)
correct answer:- 54
Question 8
The wavelength of spectral line obtained in the spectrum of $$Li^{2+}$$ ion, when the transition takes place between two levels whose sum is 4 and difference is 2, is
correct answer:- 3
Question 9
The wave numbers of three spectral lines of H atom are considered. Identify the set of spectral lines belonging to Balmer series.
(R = Rydberg constant)
correct answer:- 1
Question 10
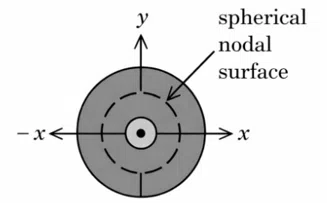
Figure 1. electron probability density for 2s orbital
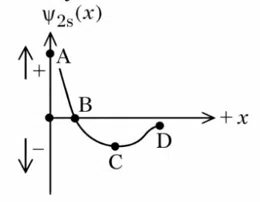
Figure 2. wave function for 2s orbital
Which of the following point in Figure 2 most accurately represents the nodal surface as shown in Figure 1?
correct answer:- 1
Question 11
The work functions of two metals ($$M_{A}$$ and $$M_{B}$$ ) are in the 1 : 2 ratio. When these metals are exposed to photons of energy 6 eV, the kinetic energy of liberated electrons of $$M_{A}$$ : $$M_{B}$$ is in the ratio of 2.642 : 1. The work functions (in eV) of $$M_{A}$$ and $$M_{A}$$ are respectively.
correct answer:- 4
Question 12
Identify the INCORRECT statements from the following:
A. Notation $$_{12}^{24}Mg$$ represents 24 protons and 12 neutrons.
B. Wavelength of a radiation of frequency $$ 4.5\times10^{15}s^{-1} $$ is $$ 6.7\times10^{-8} $$m.
C. One radiation has wavelength =$$\lambda_1(900nm) $$ and energy= $$E_{1}$$. Other radiation has wavelength = $$\lambda_2(300nm) $$ and energy= $$E2 \cdot E1 : E_2= 3 : 1$$.
D. Number of photons of light of wavelength 2000 pm that provides 1 J of
energy is $$1.006 x 10^{16}$$.
Choose the correct answer from the options given below:
correct answer:- 2
Question 13
The wavelength of photon ' A' is 400 nm. The frequency of photon ' B' is $$10^{16}s^{-1}$$. The wave number of photon 'C is $$10^{4}cm^{-1}$$.The correct order of energy of these photons is :
correct answer:- 1
Question 14
Two positively charged particles m1 and m2 have been accelerated across the same potential difference of 200 keV as shown below.
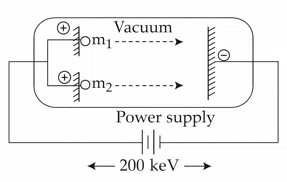
[Given mass of $$m_{1}$$ = 1amu and $$m_{2}$$ = 4amu]
The deBroglie wavelength of $$m_{1}$$ will be x times of $$m_{2}$$. The value of x is_______(nearest integer)
correct answer:- 2
Question 15
Given below are two statements : Statement (I) : A spectral line will be observed for a $$2p_{x}\rightarrow 2p_{y}$$ transition.Statement (II) :$$2p_{x}$$ and $$2p_{y}$$ are degenerate orbitals. In the light of the above statements, choose the correct answer from the options given below :
correct answer:- 2
Question 16
Radius of the first excited state of Helium ion is given as : $$a_0\rightarrow$$ radius of first stationary state of hydrogen atom.
correct answer:- 2
Question 17
Given below are two statements : Statement (I) : For a given shell, the total number of allowed orbitals is given by $$n^{2}$$. Statement (II) : For any subshell, the spatial orientation of the orbitals is given by -l to +l values including zero. In the light of the above statements, choose the correct answer from the options given below :
correct answer:- 3
Question 18
Given below are two statements about X-ray spectra of elements : Statement (I) : A plot of $$\sqrt{v}$$($$\upsilon$$ = frequency of -rays emitted) vs atomic mass is a straight line. Statement (II) : A plot of $$\upsilon(\upsilon$$ = frequency of -rays emitted) vs atomic number is a straight line. In the light of the above statements, choose the correct answer from the options given below :
correct answer:- 3
Question 19
For hydrogen atom, the orbital/s with lowest energy is/are:$$\text{ (A) } 4s \text{ (B) } 3p_x \text{ (C) } 3d_{x^2-y^2} \text{ (D) } 3d_{z^2} \text{ (E) } 4p_z$$ Choose the correct answer from the options given below:
correct answer:- 1
Question 20
In a multielectron atom, which of the following orbitals described by three quantum numbers will have same energy in absence of electric and magnetic fields? $$A. n = 1, l = 0, m_{1}=0 B.n = 2,l = 0, m_{1}=0 C.n = 2,l = 1, m_{1}=1 D.n = 3,l = 2, m_{1} = 1 E.n = 3, l = 2, m_{1} = 0$$ Choose the correct answer from the options given below:
correct answer:- 4
Question 21
If $$a_{\circ}$$ is denoted as the Bohr radius of hydrogen atom, then what is the de-Broglie wavelength $$(\lambda)$$ of the electron present in the second orbit of hydrogen atom? [n : any integer]
correct answer:- 1
Question 22
Heat treatment of muscular pain involves radiation of wavelength of about 900 nm . Which spectral line of H atom is suitable for this? Given : Rydberg constant $$R_{H}=10^{5} cm^{-1}$$, $$h=6.6 \times 10^{-34}$$ Js,$$c= 3\times 10^{8}$$ m/s
correct answer:- 3
Question 23
For hydrogen like species, which of the following graphs provides the most appropriate representation of E vs Z plot for a constant n ? [E: Energy of the stationary state, Z : atomic number, n = principal quantum number]
correct answer:- 1
Question 24
Given below are two statements : Statement (I) : It is impossible to specify simultaneously with arbitrary precision, both the linear momentum and the position of a particle. Statement (II) : If the uncertainty in the measurement of position and uncertainty in measurement of momentum are equal for an electron, then the uncertainty in the measurement of velocity is $$\ge \sqrt{\frac{h}{\pi} \times \frac{1}{2m}}$$. In the light of the above statements, choose the correct answer from the options given below :
correct answer:- 3
Question 25
The de-Broglie's wavelength of an electron in the 4th orbit is ______ $$\pi a_0$$. ($$a_0$$ = Bohr's radius)
correct answer:- 8
Question 26
Which of the following is/are not correct with respect to energy of atomic orbitals of hydrogen atom? (A)1s < 2p < 3d < 4s (B) 1s < 2s = 2p < 3s = 3p (C)1s < 2s < 2p < 3s < 3p (D) 1s < 2s < 4s < 3d
Choose the correct answer from the options given below :
correct answer:- 3
Question 27
According to the wave-particle duality of matter by de-Broglie, which of the following graph plot presents most appropriate relationship between wavelength of electron $$\lambda$$ and momentum of electron $$p$$? (Four graphs are shown: (1) $$\lambda$$ vs $$p$$ showing a rectangular hyperbola, (2) $$p$$ vs $$\lambda$$ showing a rectangular hyperbola, (3) $$\lambda$$ vs $$1/p$$ showing a straight line through origin, (4) $$\lambda$$ vs $$p$$ showing a straight line with negative slope)
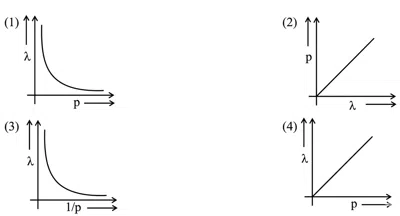
correct answer:- 1
Question 28
In case of isoelectronic species the size of $$F^-$$, $$Ne$$ and $$Na^+$$ is affected by:
correct answer:- 4
Question 29
The number of radial node/s for 3p orbital is:
correct answer:- 1
Question 30
The electronic configuration for Neodymium is: [Atomic Number for Neodymium 60]
correct answer:- 1
Question 31
The number of electrons present in all the completely filled subshells having $$n = 4$$ and $$s = +\frac{1}{2}$$ is ______ (Where $$n$$ = principal quantum number and $$s$$ = spin quantum number)
correct answer:- 16
Question 32
Total number of ions from the following with noble gas configuration is
$$Sr^{2+}$$ (Z = 38), $$Cs^+$$ (Z = 55), $$La^{2+}$$ (Z = 57), $$Pb^{2+}$$ (Z = 82), $$Yb^{2+}$$ (Z = 70) and $$Fe^{2+}$$ (Z = 26)
correct answer:- 2
Question 33
The correct set of four quantum numbers for the valence electron of rubidium atom $$(Z = 37)$$ is:
correct answer:- 1
Question 34
Given below are two statements:
Statement-I: The orbitals having same energy are called as degenerate orbitals.
Statement-II: In hydrogen atom, $$3p$$ and $$3d$$ orbitals are not degenerate orbitals.
In the light of the above statements, choose the most appropriate answer from the options given
correct answer:- 1
Question 35
Number of spectral lines obtained in $$He^+$$ spectra, when an electron makes transition from fifth excited state to first excited state will be
correct answer:- 10
Question 36
The ionization energy of sodium in $$kJ \text{ mol}^{-1}$$, if electromagnetic radiation of wavelength $$242$$ nm is just sufficient to ionize sodium atom is ______. (nearest integer)
correct answer:- 494
Question 37
The four quantum numbers for the electron in the outer most orbital of potassium (atomic no. 19) are
correct answer:- 2
Question 38
The maximum number of orbitals which can be identified with $$n = 4$$ and $$m_l = 0$$ is ______
correct answer:- 4
Question 39
In an atom, total number of electrons having quantum numbers $$n = 4$$, $$|m_l| = 1$$ and $$m_s = -\frac{1}{2}$$ is ______
correct answer:- 6
Question 40
The value of Rydberg constant $$(R_H)$$ is $$2.18 \times 10^{-18} \text{ J}$$. The velocity of electron having mass $$9.1 \times 10^{-31} \text{ kg}$$ in Bohr's first orbit of hydrogen atom $$=$$ ______ $$\times 10^5 \text{ ms}^{-1}$$ (nearest integer).
correct answer:- 22

 Group
Group






.webp)















