Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
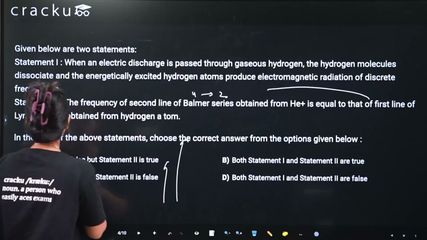
Given below are two statements:
Statement I : When an electric discharge is passed through gaseous hydrogen, the hydrogen molecules dissociate and the energetically excited hydrogen atoms produce electromagnetic radiation of discrete frequencies.
Statement II: The frequency of second line of Balmer series obtained from He+ is equal to that of first line of Lyman series obtained from hydrogen a tom.
ln the light of the above statements, choose the correct answer from the options given below :
Statement I: Electric discharge through H₂ causes dissociation and excited H atoms emit discrete frequencies. TRUE.
Statement II: 2nd line of Balmer for He⁺: $$\frac{1}{\lambda} = 4R(\frac{1}{4}-\frac{1}{16}) = 4R \times \frac{3}{16} = \frac{3R}{4}$$. 1st line of Lyman for H: $$\frac{1}{\lambda} = R(1-\frac{1}{4}) = \frac{3R}{4}$$. Equal! TRUE.
The answer is Option 2: Both true.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
