JEE General Organic Chemistry PYQs
JEE General Organic Chemistry PYQs are an important part of the JEE Chemistry syllabus. They help you understand the kind of questions asked from this chapter and check how well you know the main ideas, such as electronic effects, acidity and basicity, reaction intermediates, bond fission, hybridization, resonance, and different types of organic reactions.
In the exam, general organic chemistry questions usually come as direct concept-based questions or simple reaction-based problems. The good thing is that this chapter becomes much easier when your basics are clear. Once you understand the concepts properly and know which rule or effect to use, solving questions feels much more manageable. You do not need to think of general organic chemistry as a very difficult chapter. With regular revision and smart practice, it can become one of the more scoring parts of JEE Chemistry.
In this blog, you will find a simple formula PDF, a section for important JEE General Organic Chemistry PYQs in download format, a few practice questions with answers, and some extra questions to solve on your own. You will also learn about common mistakes students often make and a few simple tips to save time in the exam.
JEE General Organic Chemistry Important PYQs PDF
This PDF can include the most important previous year questions from general organic chemistry. It may cover topics like inductive effect, resonance, hyperconjugation, acidity, basicity, electrophiles, nucleophiles, carbocations, carbanions, free radicals, and reaction mechanisms.
Practicing these questions will help you understand the exam pattern better. It will also improve your speed, accuracy, and confidence before the exam.
Important Formulas for JEE General Organic Chemistry PYQs
You only need a few important rules, ideas, and trends to solve most general organic chemistry questions in JEE. These help you understand stability, reactivity, acidity, basicity, and reaction behaviour more clearly.
You can download the full formula PDF from the link above. Here is a quick look at some of the main rules and trends:
Concept | Formula / Rule |
Homolytic Fission | Bond breaks equally and forms free radicals |
Heterolytic Fission | Bond breaks unequally and forms ions |
Inductive Effect | Permanent electron shift through sigma bond |
Resonance Effect | Delocalization of electrons in a molecule |
Hyperconjugation | Delocalization involving alpha hydrogen |
Stability of Carbocation | 3° > 2° > 1° > CH₃⁺ |
Stability of Carbanion | CH₃⁻ > 1° > 2° > 3° |
Stability of Free Radical | 3° > 2° > 1° > CH₃• |
Acidity Rule | Greater stability of conjugate base means stronger acid |
Basicity Rule | Greater availability of lone pair means stronger base |
These formulas and rules are commonly used in questions based on electronic effects, acidity, basicity, intermediates, and organic reaction mechanisms. If you revise them properly, many JEE questions will start to feel much easier.
Top 5 Common Mistakes to Avoid in JEE General Organic Chemistry PYQs
Many students find general organic chemistry confusing at first because it includes many concepts and trends. But most mistakes happen because small details are missed while solving. Here are some common mistakes you should avoid:
Mixing up inductive effect and resonance effect
Both affect electron movement, but they are not the same. Inductive effect works through sigma bonds, while resonance works through pi bonds or lone pairs.
Confusing electrophiles and nucleophiles
Electrophiles accept electrons, while nucleophiles donate electrons. Many students mix them up in reaction-based questions.
Forgetting the correct order of intermediate stability
Carbocations, carbanions, and free radicals do not all follow the same stability trend. If you do not revise these properly, even easy questions can become confusing.
Ignoring the role of resonance in acidity and basicity
The strength of an acid or base often depends on the stability of the conjugate species. Students sometimes focus on only one factor and miss the full concept.
Not reading the reaction condition carefully
In organic chemistry, a small change in reagent or condition can change the product completely. Always read the question carefully before answering.
List of JEE General Organic Chemistry PYQs
Here is a short set of JEE-style general organic chemistry questions for practice. These include common question types from electronic effects, acidity, basicity, intermediates, and reaction concepts. Solving them regularly can help you become faster and more confident.
Question 1
Given below are four compounds :
(a) n-propyl choride
(b) iso-propyl chloride
(c) sec-butyl chloride
(d) neo-pentyl chloride
Percentage of carbon in the one which exhibits optical isomerism is:
correct answer:- 2
Question 2
Match List - I with List - II.

Choose the correct answer from the options given below :
correct answer:- 1
Question 3
From the following, the least stable structure is :
correct answer:- 2
Question 4
80 mL of a hydrocarbon on mixing with 264 mL of oxygen in a closed U-tube undergoes complete combustion. The residual gases after cooling to 273 K occupy 224 mL. When the system is treated with KOH solution, the volume decreases to 64 ml. The formula of the hydrocarbon is:
correct answer:- 1
Question 5
Identify correct statements from the following :
A Propanal and propanone are functional isomers.
B. Ethoxyethane and methoxypropane are metamers.
C. But-2-ene shows optical isomerism.
D. But-1-ene and but-2-ene are functional isomers.
E. Pentane and 2, 2-dimethyl propane ai-e chain isomers.
Choose the correct answer from the options given below:
correct answer:- 2
Question 6
The IUPAC name of the following compound is :
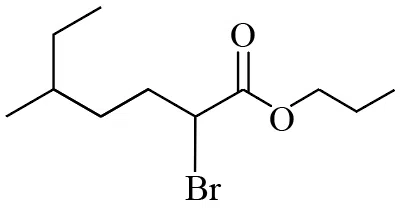
correct answer:- 2
Question 7
Arrange the following carbanions in the decreasing order of stability.
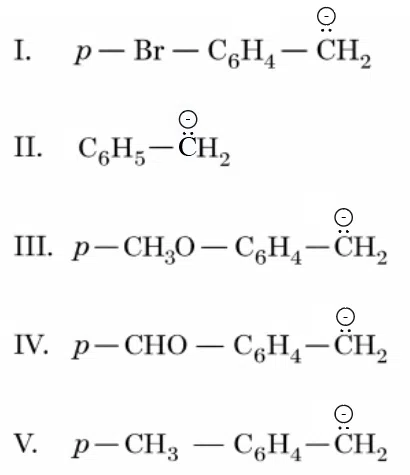
Choose the correct answer from the options given below:
correct answer:- 4
Question 8
Given below are two statements:
Statement I: There are several conformers for n-butane. Out of those conformers,
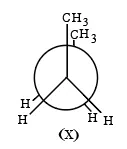
is the least stable and most stable conformer is
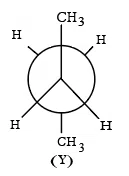
Statement II: As the dihedral angle increases, torsional strain decreases from (X) to (Y).
In the light of the above statements, choose the correct answer from the options given below
correct answer:- 2
Question 9
0.53 g of an organic compound (x) when heated with excess of nitric acid ( concentrated) and then with silver nitrate gave 0. 75 g of silver bromide precipitate. 1.0 g of (x) gave 1.32 g of $$CO_{2}$$ gas on combustion. The percentage of hydrogen in the compound (x) is __ %. [Nearest Integer]
[Given: Molar mass in g $$mol^{-1}$$ H : 1, C : 12, Br: 80, Ag: 108, O : 16 ; Compound (x) :$$C_{x}H_{y}Br_{z}$$]
correct answer:- 4
Question 10
Given below are two statements:
Statement I: $$(CH_3)_3C^+$$ is more stable than $$CH_3^+$$ as nine hyperconjugation interactions are possible in $$(CH_3)_3C^+$$.
Statement II: $$CH_3^+$$ is less stable than $$(CH_3)_3C^+$$ as only three hyperconjugation interactions are possible in $$CH_3^+$$.
In the light of the above statements, choose the correct answer from the options given below
correct answer:- 3
Question 11
The cyclic cations having the same number of hyperconjugalion are :
A.
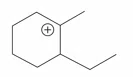
B.
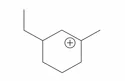
C.
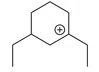
D.
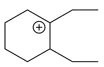
Choose the correct answer from the options given below :
correct answer:- 3
Question 12
The most stable carbocation from the following is :
correct answer:- 3
Question 13
How many different stereoisomers are possible for the given molecule?
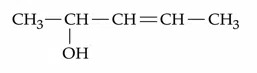
correct answer:- 3
Question 14
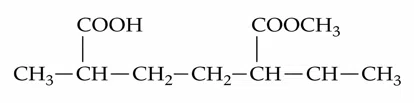
The IUPAC name of the following compound is :
correct answer:- 3
Question 15
In Carius method for estimation of halogens, 180 mg of an organic compound produced 143.5 mg of AgCl . The percentage composition of chlorine in the compound is _______ \%. (Given : molar mass in $$gmol^{-1} \text{ of } Ag: 108, Cl: 35.5$$)
correct answer:- 20
Question 16
Which one of the carbocations from the following is most stable?
correct answer:- 2
Question 17
$$\text{Identify correct statement/s : }(A) -OCH_3 \text{ and } -NHCOCH_3 \text{ are activating groups. } (B)-CN \text{ and } -OH \text{ are meta directing groups. } (C) -CN \text{ and } -SO_3H$$ are meta directing groups. $$(D)$$ Activating groups act as ortho- and para-directing groups. $$(E)$$ Halides are activating groups. Choose the correct answer from the options given below:
correct answer:- 4
Question 18
In the given structure, the number of $$sp \text{ and } sp^2 $$ hybridized carbon atoms present respectively are:
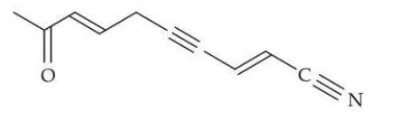
correct answer:- 2
Question 19
$$\text{The possible number of stereoisomers for 5-phenylpent -4-en-2-ol is } \underline{\hspace{2cm}}.$$
correct answer:- 4
Question 20
The hydrocarbon $$(X)$$ with molar mass $$80\,g\,mol^{-1}$$ and $$90\% $$ carbon has $$\underline{\hspace{2cm}}$$ degree of unsaturation.
correct answer:- 3
Question 21
The correct order of stability of following carbocations is :
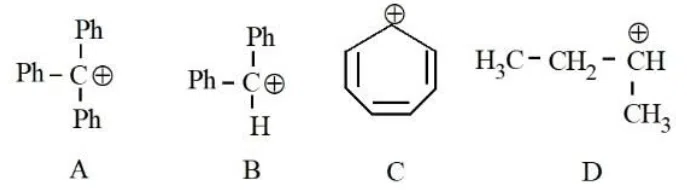
correct answer:- 4
Question 22
Total number of nucleophiles from the following is :
$$NH_{3},PhSH,(H_{3}C)_{2}S,H_{2}C=CH_{2},\ominus\\O H,H_{3}O^{\oplus},(CH_{3})_{2}CO,\rightleftharpoons NCH_{3}$$
correct answer:- 4
Question 23
Match List - I with List - II.
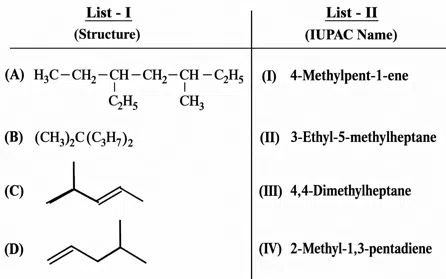
Choose the correct answer from the options given below :
correct answer:- 1
Question 24
The correct stability order of the following species/molecules is:
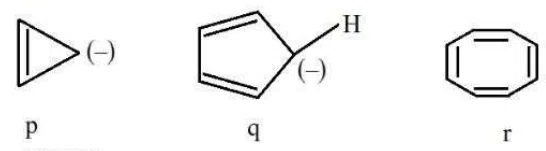
correct answer:- 1
Question 25
Match List I with List II :
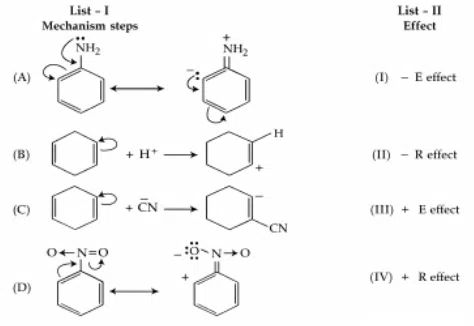
Choose the correct answer from the options given below :
correct answer:- 1
Question 26
Which among the following is incorrect statement?
correct answer:- 3
Question 27
Given below are two statements : Statement (I) :
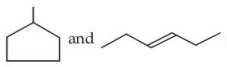
are isomeric compounds. Statement (II) :
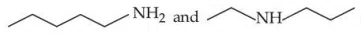
are functional group isomers.
correct answer:- 2
Question 28
Ionic reactions with organic compounds proceed through: (A) Homolytic bond cleavage (B) Heterolytic bond cleavage (C) Free radical formation (D) Primary free radical (E) Secondary free radical. Choose the correct answer from the options given below:
correct answer:- 3
Question 29
Number of optical isomers possible for 2-chlorobutane is:
correct answer:- 2
Question 30
The functional group that shows negative resonance effect is:
correct answer:- 3
Question 31
IUPAC name of the following compound (P) is :
correct answer:- 2
Question 32
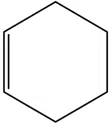
Cyclohexene is _________ type of an organic compound.
correct answer:- 4
Question 33
Bond line formula of $$HOCH(CN)_2$$ is :
correct answer:- 4
Question 34
The order of relative stability of the contributing structures is :
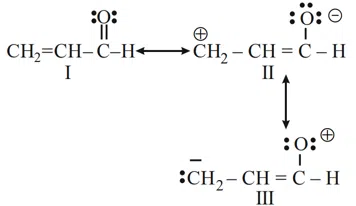
Choose the correct answer from the options given below:
correct answer:- 1
Question 35
The incorrect statement regarding conformations of ethane is :
correct answer:- 3
Question 36
Total number of compounds with Chiral carbon atoms from following is
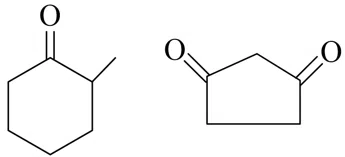
$$CH_3-CH_2-CHNO_2-COOH$$
$$CH_3-CH(I)-CH_2-NO_2$$
$$CH_3-CH_2-CHBr-CH_2-CH_3$$
$$CH_3-CH_2-CH(OH)-CH_2OH$$
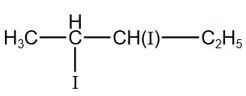
correct answer:- 5
Question 37
The interaction between $$\pi$$ bond and lone pair of electrons present on an adjacent atom is responsible for
correct answer:- 4
Question 38
The difference in energy between the actual structure and the lowest energy resonance structure for the given compound is:
correct answer:- 2
Question 39
Number of compounds among the following which contain sulphur as heteroatom is _______.
Furan, Thiophene, Pyridine, Pyrrole, Cysteine, Tyrosine
correct answer:- 2
Question 40
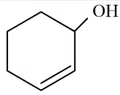
According to IUPAC system, the compound is named as:
correct answer:- 4
Question 41
The ascending acidity order of the following H atoms is
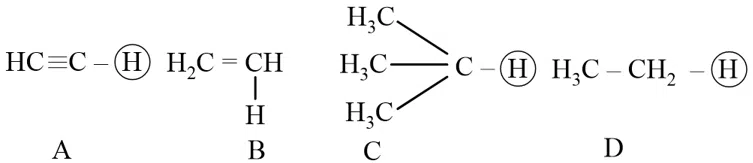
correct answer:- 1
Question 42
Match List I with List II
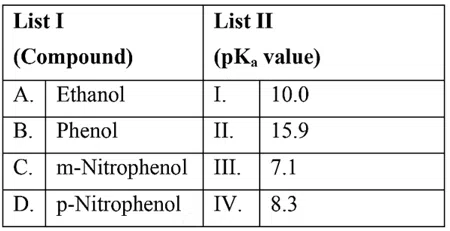
Choose the correct answer from the options given below:
correct answer:- 4
Question 43
Which one of the following will show geometrical isomerism?
correct answer:- 2
Question 44
The total number of 'Sigma' and Pi bonds in 2-formylhex-4-enoic acid is ______.
correct answer:- 22
Question 45
Which of the following molecule/species is most stable?
correct answer:- 1
Question 46
IUPAC name of the following compound is:
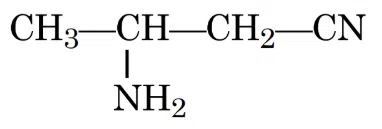
correct answer:- 3
Question 47
The correct stability order of carbocations is
correct answer:- 3
Question 48
Number of geometrical isomers possible for the given structure is/are ________.
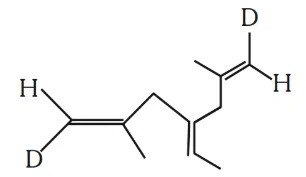
correct answer:- 4
Question 49
2-chlorobutane + $$Cl_2 \rightarrow C_4H_8Cl_2$$ (isomers)
Total number of optically active isomers shown by $$C_4H_8Cl_2$$, obtained in the above reaction is ________.
correct answer:- 6
Question 50
A species having carbon with sextet of electrons and can act as electrophile is called
correct answer:- 3

 Group
Group






.webp)













