Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
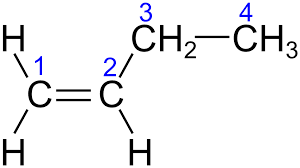
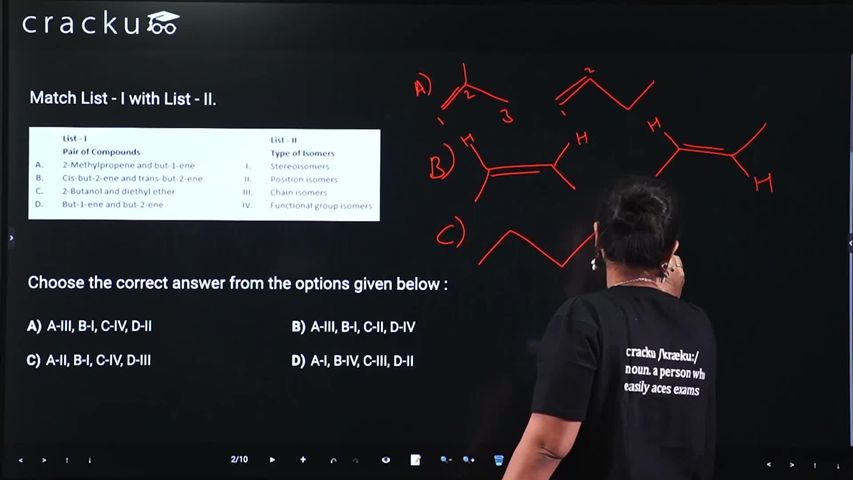
Match List - I with List - II.

Choose the correct answer from the options given below :
A) Chain isomer - Both have the formula $$(C_{4}H_{8})$$ but different carbon skeletons (branched vs. straight chain).
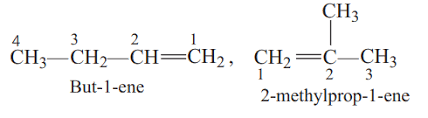
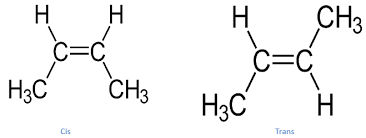
B)Stereo isomers - They have the same connectivity but differ in the spatial arrangement of atoms around the double bond.
C)Functional group isomers - Both have the formula $$(C_4H_{10}O)$$ but belong to different functional classes (alcohol vs. ether).
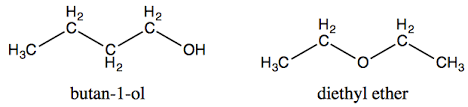
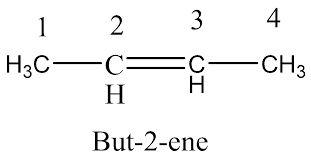
D)Position isomers - They have the same carbon chain but the double bond is at different positions (C1 vs. C2).
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
