Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
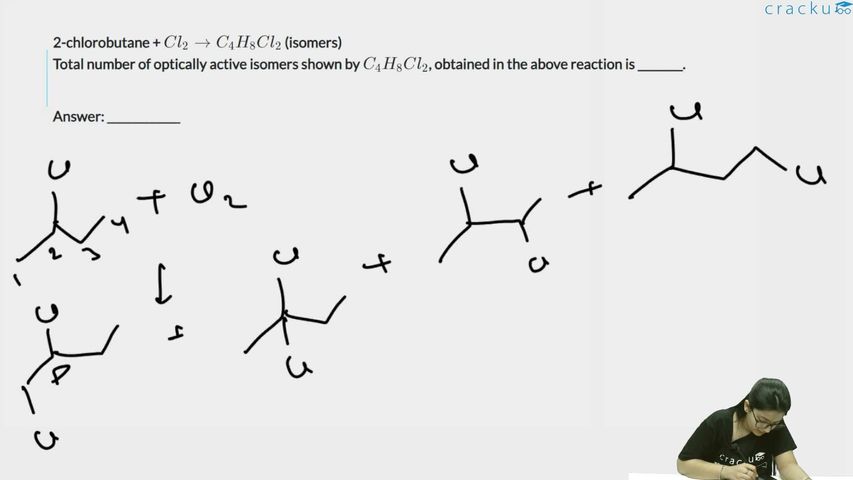
2-chlorobutane + $$Cl_2 \rightarrow C_4H_8Cl_2$$ (isomers)
Total number of optically active isomers shown by $$C_4H_8Cl_2$$, obtained in the above reaction is ________.
Correct Answer: 6
2-chlorobutane: CH$$_3$$CHClCH$$_2$$CH$$_3$$. Monochlorination gives C$$_4$$H$$_8$$Cl$$_2$$ isomers.
Possible positions for the second Cl:
1. C1: CH$$_2$$ClCHClCH$$_2$$CH$$_3$$ — C2 is chiral, optically active (2 enantiomers)
2. C2: CH$$_3$$CCl$$_2$$CH$$_2$$CH$$_3$$ — no chiral center, not optically active
3. C3: CH$$_3$$CHClCHClCH$$_3$$ — two chiral centers, can have (R,R), (S,S), and meso. The (R,R) and (S,S) are optically active = 2 enantiomers. Also (R,S) which is meso.
4. C4: CH$$_3$$CHClCH$$_2$$CH$$_2$$Cl — C2 is chiral, 2 enantiomers
Total optically active isomers: 2 + 0 + 2 + 2 = 6.
Therefore, the answer is $$\boxed{6}$$.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
