Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The total number of 'Sigma' and Pi bonds in 2-formylhex-4-enoic acid is ______.
Correct Answer: 22
We need to find the total number of sigma ($$\sigma$$) and pi ($$\pi$$) bonds in 2-formylhex-4-enoic acid.
2-formylhex-4-enoic acid has a 6-carbon chain (hexanoic acid) with a formyl group ($$-CHO$$) at C2 and a double bond between C4 and C5.
Structure: $$CH_3-CH=CH-CH_2-CH(CHO)-COOH$$
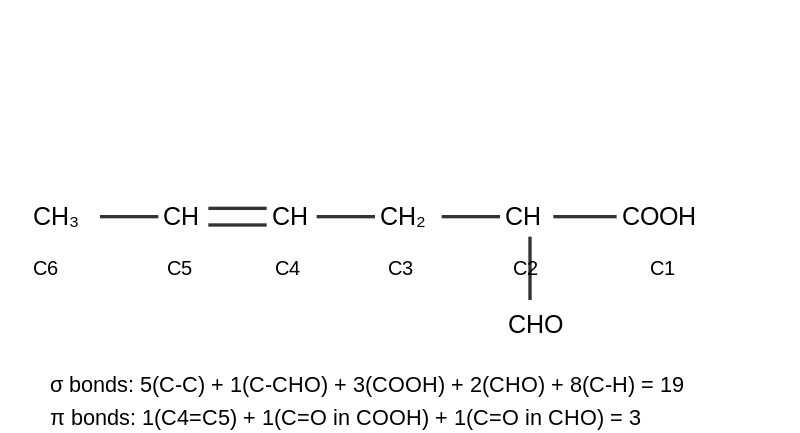
Every single bond is 1$$\sigma$$ bond, and every double bond contains 1$$\sigma$$ + 1$$\pi$$ bond.
C-C sigma bonds in main chain: C1-C2, C2-C3, C3-C4, C4=C5($$\sigma$$), C5-C6 = 5
C2-CHO branch: 1
In $$-COOH$$: C=O($$\sigma$$) + C-OH + O-H = 3
In $$-CHO$$: C=O($$\sigma$$) + C-H = 2
C-H bonds: C2(1H) + C3(2H) + C4(1H) + C5(1H) + C6(3H) = 8
Total $$\sigma$$ bonds = 5 + 1 + 3 + 2 + 8 = 19
C4=C5: 1$$\pi$$ bond
C=O in $$-COOH$$: 1$$\pi$$ bond
C=O in $$-CHO$$: 1$$\pi$$ bond
Total $$\pi$$ bonds = 3
Total = 19 + 3 = 22
The total number of sigma and pi bonds is $$\boxed{22}$$.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
