Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
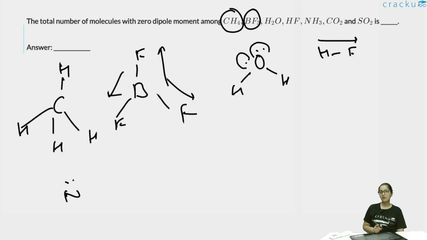
The total number of molecules with zero dipole moment among $$CH_4, BF_3, H_2O, HF, NH_3, CO_2$$ and $$SO_2$$ is ______.
Correct Answer: 3
We need to find the number of molecules with zero dipole moment among: $$CH_4, BF_3, H_2O, HF, NH_3, CO_2,$$ and $$SO_2$$.
A molecule has zero dipole moment when the individual bond dipoles cancel each other out due to symmetry.
Analysing each molecule:
1. $$CH_4$$ (Methane): Tetrahedral geometry. All four $$C-H$$ bonds are identical and symmetrically arranged. The bond dipoles cancel out completely.
Dipole moment = Zero
2. $$BF_3$$ (Boron trifluoride): Trigonal planar geometry. The three $$B-F$$ bonds are symmetrically placed at $$120°$$ angles. The bond dipoles cancel out.
Dipole moment = Zero
3. $$H_2O$$ (Water): Bent geometry with a bond angle of about $$104.5°$$. The two $$O-H$$ bond dipoles do not cancel due to the bent shape, and there are two lone pairs on oxygen.
Dipole moment = Non-zero
4. $$HF$$ (Hydrogen fluoride): Linear diatomic molecule with a highly polar $$H-F$$ bond. Since there is only one bond, there is nothing to cancel it.
Dipole moment = Non-zero
5. $$NH_3$$ (Ammonia): Trigonal pyramidal geometry with one lone pair on nitrogen. The bond dipoles and the lone pair contribution do not cancel.
Dipole moment = Non-zero
6. $$CO_2$$ (Carbon dioxide): Linear geometry. The two $$C=O$$ bonds are equal and opposite in direction. The bond dipoles cancel perfectly.
Dipole moment = Zero
7. $$SO_2$$ (Sulfur dioxide): Bent geometry with a bond angle of about $$119°$$. The two $$S=O$$ bond dipoles do not cancel due to the bent shape, and there is a lone pair on sulfur.
Dipole moment = Non-zero
The molecules with zero dipole moment are: $$CH_4$$, $$BF_3$$, and $$CO_2$$.
Therefore, the total number of molecules with zero dipole moment = 3.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
