Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
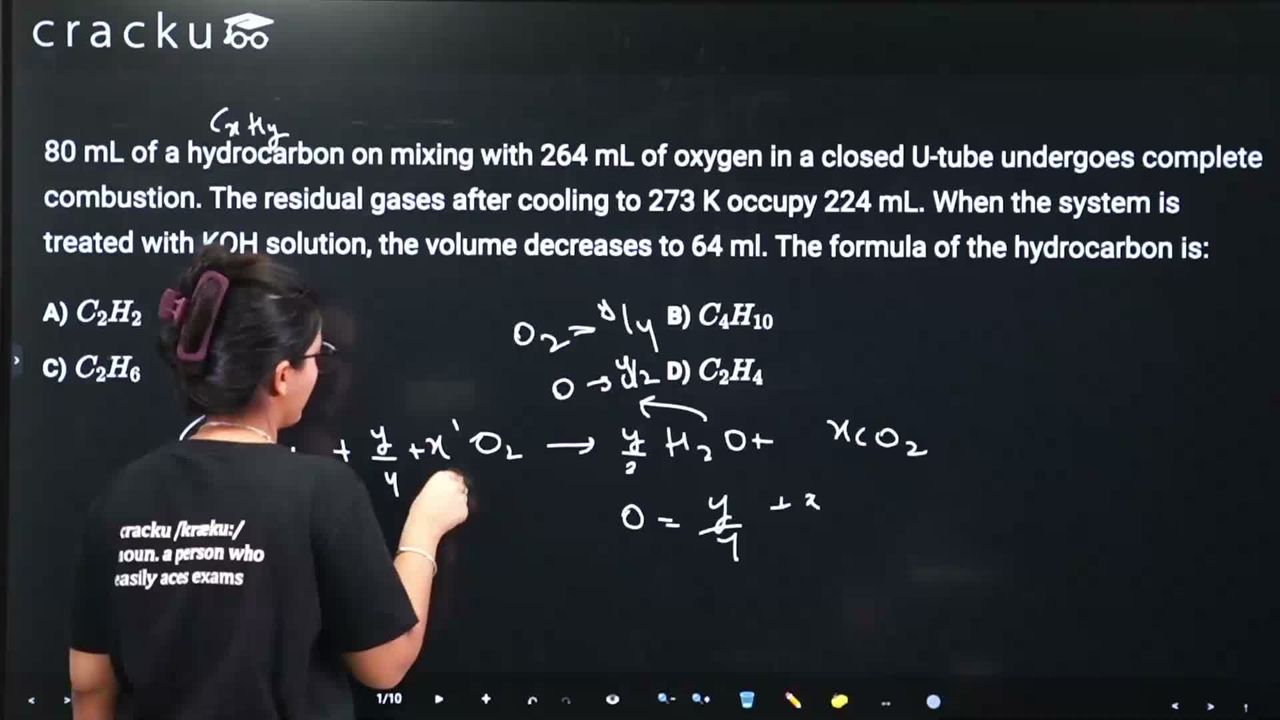
80 mL of a hydrocarbon on mixing with 264 mL of oxygen in a closed U-tube undergoes complete combustion. The residual gases after cooling to 273 K occupy 224 mL. When the system is treated with KOH solution, the volume decreases to 64 ml. The formula of the hydrocarbon is:
80 mL hydrocarbon + 264 mL O₂. After combustion at 273K: 224 mL residual (CO₂ + unreacted O₂). After KOH: 64 mL (unreacted O₂, since KOH absorbs CO₂).
CO₂ volume = 224 - 64 = 160 mL. O₂ used = 264 - 64 = 200 mL.
$$C_xH_y + (x + y/4)O_2 \to xCO_2 + y/2 H_2O$$
From 80 mL: $$80x = 160 \Rightarrow x = 2$$. $$80(x+y/4) = 200 \Rightarrow 2+y/4 = 2.5 \Rightarrow y = 2$$.
Formula: $$C_2H_2$$ (acetylene).
The answer is Option 1: $$C_2H_2$$.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
