Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
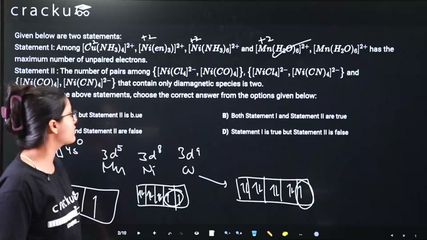
Given below are two statements:
Statement I: Among $$[Cu(NH_{3})_{4}]^{2+},[Ni(en)_{3})]^{2+},[Ni(NH_{3})_{6}]^{2+}$$ and $$[Mn(H_{2}O)_{6}]^{2+},[Mn(H_{2}O)_{6}]^{2+}$$ has the maximum number of unpaired electrons.
Statement II : The number of pairs among $$\left\{[Ni(Cl_{4}]^{2-},[Ni(CO)_{4}]\right\}$$, $$\left\{[NiCl_{4}]^{2-},[Ni(CN)_{4}]^{2-}\right\}$$ and $$\left\{[Ni(CO)_{4}],[Ni(CN)_{4}]^{2-}\right\}$$ that contain only diamagnetic species is two.
ln the light of the above statements, choose the correct answer from the options given below:
Statement I: Unpaired electrons: [Cu(NH₃)₄]²⁺: Cu²⁺ d⁹, strong field → 1; [Ni(en)₃]²⁺: Ni²⁺ d⁸, strong field → 2; [Ni(NH₃)₆]²⁺: Ni²⁺ d⁸, weak field → 2; [Mn(H₂O)₆]²⁺: Mn²⁺ d⁵, weak field → 5. Maximum is Mn with 5. TRUE.
Statement II: [NiCl₄]²⁻: tetrahedral, Ni²⁺ d⁸, 2 unpaired → paramagnetic. [Ni(CO)₄]: Ni⁰ d¹⁰ → diamagnetic. [Ni(CN)₄]²⁻: square planar, Ni²⁺ d⁸ → diamagnetic. Pairs with only diamagnetic: {[Ni(CO)₄],[Ni(CN)₄]²⁻} → 1 pair. Statement says 2. FALSE.
The answer is Option 4: Statement I is true but Statement II is false.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
