JEE Aldehydes, Ketones & Carboxylic Acids PYQs
The JEE Chemistry of Aldehydes, Ketones and Carboxylic Acids PYQs is significant. They make you know the type of questions that are asked in this chapter and how familiar you are to the key topics, which are structure, naming, methods of preparation, reactions, acidity, nucleophilic addition, and reactions that are important in names.
Questions on this chapter in the exam often occur in two forms. Others are concept based questions but are direct, and others are reaction based problems. The good thing is that this chapter is far easier when you have your basics well in place. Once you have a clear understanding of the concepts and are aware of which reaction or rule to apply, then most questions are easier to solve. You should not consider this chapter too hard. It can become a scoring section of JEE Chemistry with frequent revision and with practice.
In this blog, you will find a simple formula PDF, a section for important JEE Aldehydes, Ketones & Carboxylic Acids PYQs in download format, a few practice questions with answers, and some extra questions to solve on your own. You will also go through common mistakes students make and a few simple tips that can help you save time in the exam.
JEE Aldehydes, Ketones & Carboxylic Acids Important PYQs PDF
This PDF may consist of the most significant past-year questions of aldehydes, ketones, and carboxylic acids. It can address such issues as nomenclature, preparation, physical properties, nucleophilic addition reactions, oxidation and reduction reactions, acidity of carboxylic acids, and name reactions of significance.
These questions will assist you in knowing the pattern of the exams better. It will also make you faster, more accurate and confident prior to the exam.
Important Formulas for JEE Aldehydes, Ketones & Carboxylic Acids PYQs
You only need a few important formulas, rules, and reactions to solve most questions from aldehydes, ketones, and carboxylic acids in JEE. These help you understand structure, reactivity, acidity, and product formation in a much clearer way.
You can download the full formula PDF from the link above. Here is a quick look at some of the main formulas and rules:
Concept | Formula / Rule |
General Formula of Aldehyde | RCHO |
General Formula of Ketone | RCOR′ |
General Formula of Carboxylic Acid | RCOOH |
Oxidation of Aldehyde | Aldehyde easily oxidizes to carboxylic acid |
Reduction of Aldehyde | Aldehyde reduces to primary alcohol |
Reduction of Ketone | Ketone reduces to secondary alcohol |
Carboxylic Acid + Alcohol | Ester + Water |
Tollen’s Test | Aldehydes give silver mirror |
Fehling’s Test | Aliphatic aldehydes give red precipitate |
Acidity Rule | Greater stability of carboxylate ion means stronger acid |
These formulas and rules are commonly used in questions based on reactions, structure, acidity, tests, and preparation methods. If you revise them well, many JEE questions will start to feel much simpler.
Top 5 Common Mistakes to Avoid in JEE Aldehydes, Ketones & Carboxylic Acids PYQs
Many students find this chapter confusing in the beginning because it has many reactions and many compounds that look similar. But in most cases, students lose marks because of small mistakes. Here are some common ones you should avoid:
Confusing aldehydes and ketones
Both contain the carbonyl group, but the position is different. In aldehydes, the carbonyl group is at the end of the carbon chain. In ketones, it is present in the middle. This small difference changes their behaviour.
Forgetting which compound gives which test
Students often mix up Tollen’s test, Fehling’s test, and Schiff’s test. You should clearly remember which functional group responds to which test.
Mixing up oxidation and reduction products
Aldehydes, ketones, and carboxylic acids do not react in the same way during oxidation and reduction. If you do not revise these reactions properly, it is easy to make mistakes.
Ignoring the effect of electron-withdrawing groups on acidity
The acidity of carboxylic acids changes when electron-withdrawing groups are attached. Many students forget this while comparing acidic strength.
Not reading the reaction conditions carefully
In organic chemistry, reagent and condition matter a lot. A small change in catalyst, medium, or reagent can give a different product.
List of JEE Aldehydes, Ketones & Carboxylic Acids PYQs
Here is a short set of JEE-style questions for practice. These cover common question types from structure, reactions, acidity, functional group tests, and name reactions. Solving them regularly can help you become faster and more confident.
Question 1
Given below are two statements:
Statement I: Compound (X), shown below, dissolves in $$NaHCO_{3}$$ solution and has two chiral carbon atoms
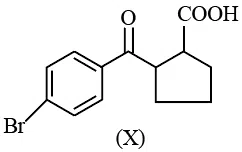
Statement II: Compound (Y), shown below, has two carbons with $$sp^{3}$$ hybridization, one carbon with $$sp^{2}$$ and one carbon with sp hybridization
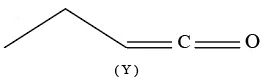
In the light of the above statements, choose the correct answer from the options given below:
correct answer:- 1
Question 2
Match List - I with List - II.

Choose the correct answer from the options given below:
correct answer:- 3
Question 3
An organic compound "P" of molecular formula $$C_{6}H_{12}O_{3}$$ gives positive Iodoform test but negative Tollen's test. When "P" is treated with dilute acid, it produces "Q". "Q" gives positive Tollen's test and also iodoform test. The structure of "P" is :
correct answer:- 2
Question 4
The compound A, $$C_{8}H_{8}O_{2}$$ reacts with acetophenone to form a single product via cross-Aldol condensation. The compound A on reaction with cone. NaOH forms a substituted benzyl alcohol as one of the two products. The compound A is :
correct answer:- 4
Question 5
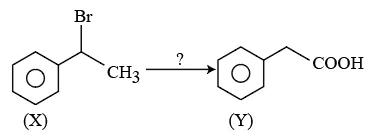
The correct sequence of reagents for the above conversion of X to Y is:
correct answer:- 2
Question 6
Compound 'P' undergoes the following sequence of reactions:
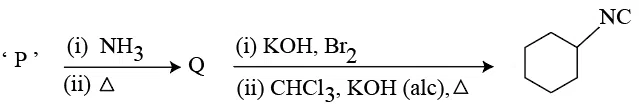
'P' is:
correct answer:- 1
Question 7
From the given following (A to D) cyclic structures, those which will not react with Tollen's reagent are:
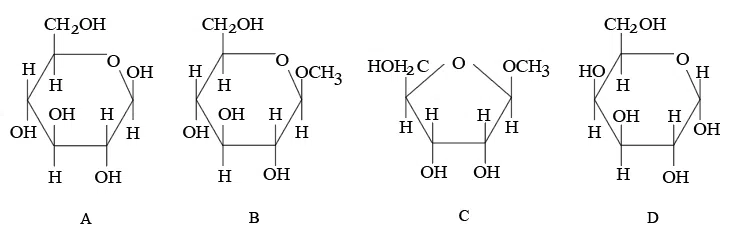
correct answer:- 3
Question 8
'x' is the product which is obtained &om; propanenitrile and stannous chloride in the presence of hydrochloric acid followed by hydrolysis. 'y' is the product which is obtained from the but-2-ene by the ozonolysis followed by hydrolysis. From the followu1g, which product is not obtained when one mole of 'x' and one mole of 'y' react with, each other in the presence of alkali followed by heating?
correct answer:- 2
Question 9
Given below are two statements:
Statement I: Phenol on treatment with . $$CHCL_{3}$$/aq. $$KOH$$ under refluxing condition, followed by acidification produces $$p$$-hydroxy benzaldehyde as the major product and $$o$$-hydroxy benzaldehyde as the minor product.
Statement II: The mixture of $$p$$-hydroxybenzaldehyde and $$o$$-
hydroxybenzaldehyde can be easily separated through steam distillation.
In the light of the above statements, choose the correct answer from the options given below
correct answer:- 1
Question 10
Match the LIST-I with LIST-II
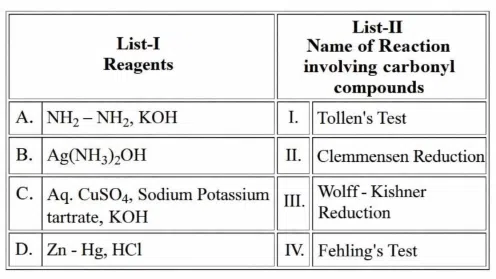
Choose the correct answer from the options given below:
correct answer:- 4
Question 11
A student is given one compound among the following compounds that gives positive test with Tollen's reagent
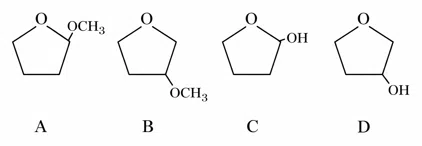
The compound is :
correct answer:- 2
Question 12
Given below are two statements:
Statement I: Cross aldol condensation between two different aldehydes will always produce four different products.
Statement II: When semicarbazide reacts with a mixture of benzaldehyde and acetophenone under optimum pH, it fonns a condensation product with acetophenone only.
In the light of the above statements, choose the correct answer from the options given below
correct answer:- 2
Question 13
Grignard reagent RMgBr (P) reacts with water and forms a gas (Q). One gram of Q occupies $$1.4 dm^{3}$$ at STP.(P) on reaction with dry ice in dry ether followed by $$H_{3}O^{+}$$ fonns a compound (Z). 0.1 mole of(Z) will weigh ____ g. (Nearest integer)
correct answer:- 6
Question 14
Given below are two statements for the following reaction sequence.
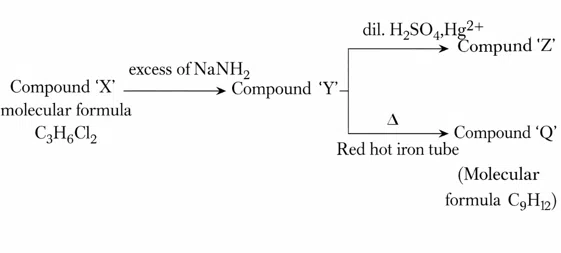
Statement I: Compound 'Z' will give yellow precipitate with NaOI.
Statement II: Compound 'Q' has two different types of'H' atoms (aromatic:aliphatic) in the ratio 1 :3.
In the light of the above statements, choose the correct answer from the options given below:
correct answer:- 4
Question 15
Given below are the four isomeric compounds (P, Q, R, S)
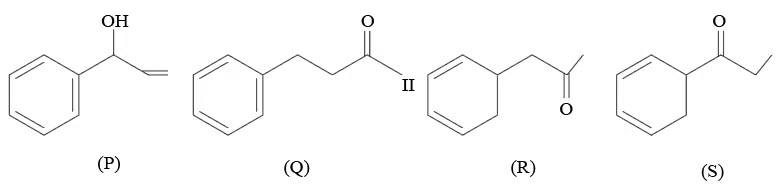
Identify correct statements from below
A. Q, R and S will give precipitate with 2, 4 - DNP.
B. P and Q will give positive Bayer's test.
C. Q and R will give sooty flame.
D. Rand S will give yellow precipitate with $$I_{2}/NaOH$$.
E. Q alone will deposit silver with Tollen's reagent.
Choose the correct option.
correct answer:- 3
Question 16
Iodoform test can differentiate between
A. Methanol and Ethanol
B. $$CH_{3}COOH$$ and $$CH_{3}CH_{2}COOH$$
C. Cyclohexene and cyclohexanone
D. Diethyl ether and Pentan-3-one
E. Anisole and acetone
Choose the correct answer from the options given below:
correct answer:- 3
Question 17
Which of the following statements are TRUE about Haloform reaction?:
A. Sodium hypochlorite reacts with KI to give KOI.
B. KOI is a reducing agent.
C. $$ \alpha,\beta$$-unsaturated methylketone $$\mathrm{CH_3 - CH = CH - C(=O) - CH_3}$$ will give iodoform reaction.
D. Isopropyl alcohol will not give iodoform test.
E. Methanoic acid will give positive iodoform test.
Choose the correct answer from the options given below:
correct answer:- 4
Question 18
The correct order of acidic strength of the major products formed in the given reactions, is:
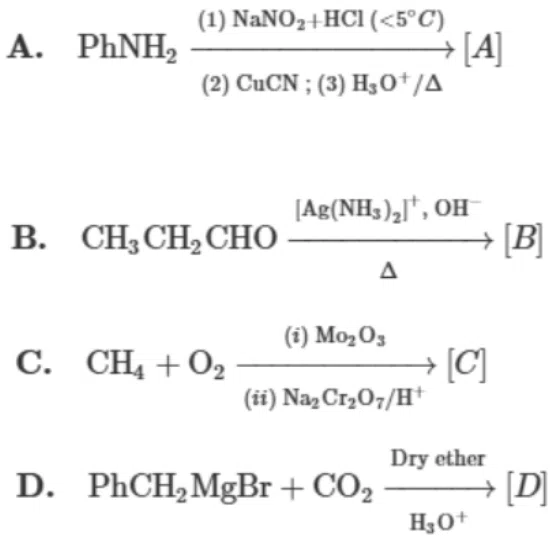
Choose the correct answer from the options given below :
correct answer:- 4
Question 19
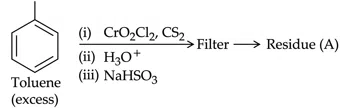
Residue (A) + HCl (dil)$$\rightarrow $$ Compound (B) Structure of residue (A) and Compound (B) formed respectively is :
correct answer:- 4
Question 20
The compounds which give positive Fehling's test are :
(A).
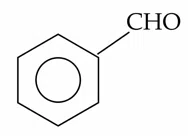
(B).
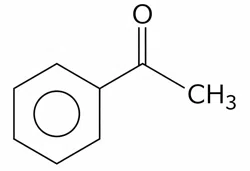
(C). $$ HOCH_{2} - CO - (CHOH)_{3}-CH_{2}-OH $$
(D).
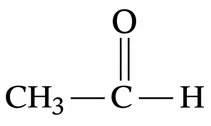
(E).
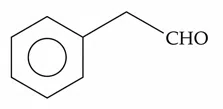
Choose the correct answer from the options given below :
correct answer:- 2
Question 21
Given below are two statements : Consider the following reaction
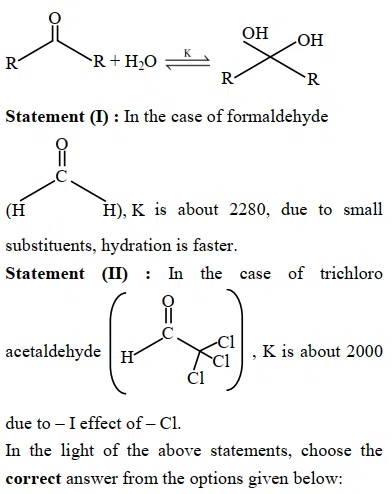
correct answer:- 4
Question 22
A compound 'X' absorbs 2 moles of hydrogen and 'X' upon oxidation with $$KMnO_{4}|H^{+}$$ gives

The total number of $$\sigma$$ bonds present in the compound 'X' is ________.
correct answer:- 27
Question 23
Aman has been asked to synthesise the molecule
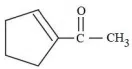
(x).He thought of preparing the molecule using an aldol condensation reaction. He found a few cyclic alkenes in his laboratory. He thought of performing ozonolysis reaction on alkene to produce a dicarbonyl compound followed by aldol reaction to prepare " x ". Predict the suitable alkene that can lead to the formation of " x ".
correct answer:- 3
Question 24
Which of the following arrangements with respect to their reactivity in nucleophilic addition reaction is correct?
correct answer:- 4
Question 25
Both acetaldehyde and acetone (individually) undergo which of the following reactions? A. Iodoform Reaction B. Cannizaro Reaction C. Aldol Condensation D. Tollen's Test E. Clemmensen Reduction Choose the correct answer from the options given below:
correct answer:- 3
Question 26
A molecule (" P ") on treatment with acid undergoes rearrangement and gives ("Q"). ("Q") on ozonolysis followed by reflux under alkaline condition gives (" R "). The structure of (" R ") is given below.
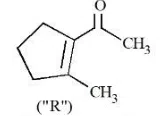
The structure of("P")is
correct answer:- 4
Question 27
The compounds that produce $$CO_{2}$$ with aqueous $$NaHCO_{3}$$ solution are:
A.
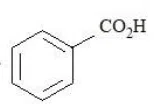
B.
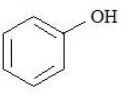
C.
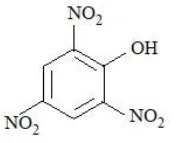
D.
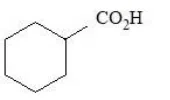
E.
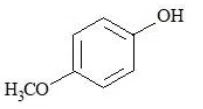
Choose the correct answer from the options given below:
correct answer:- 1
Question 28
The product (P) formed in the following reaction is :
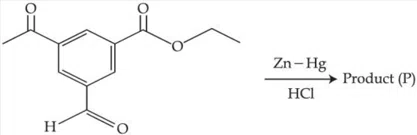
correct answer:- 1
Question 29
The major product of the following reaction is:
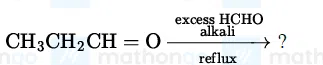
correct answer:- 1
Question 30
In the Claisen-Schmidt reaction to prepare, dibenzalacetone from 5.3 g of benzaldehyde, a total of 3.51 g of product was obtained. The percentage yield in this reaction was ______ %.
correct answer:- 60
Question 31
Given below are two statements: Statement I: Acidity of α-hydrogens of aldehydes and ketones is responsible for Aldol reaction. Statement II: Reaction between benzaldehyde and ethanal will NOT give Cross-Aldol product. Choose the most appropriate answer from the options given below :
correct answer:- 1
Question 32
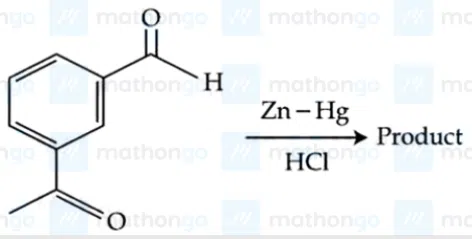
Identify the product in the following reaction :
correct answer:- 1
Question 33
The major product of the following reaction is :
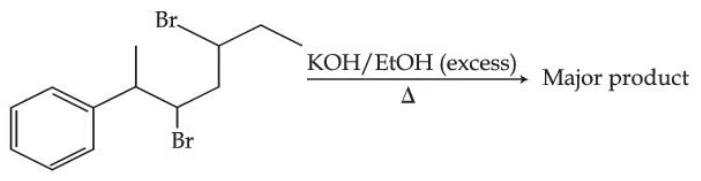
correct answer:- 4
Question 34
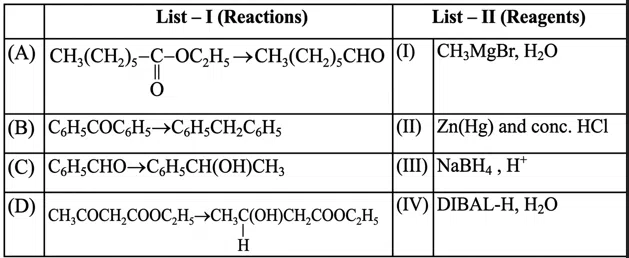
Choose the correct answer from options given below:
correct answer:- 2
Question 35
Highest enol content will be shown by :
correct answer:- 2
Question 36
Identify A in the following reaction.
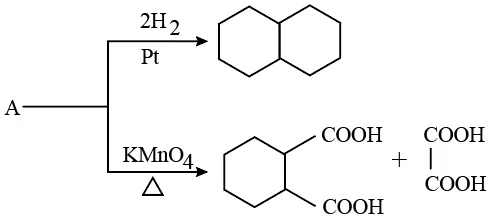
correct answer:- 4
Question 37
Match List - I with List - II.

choose the correct answer from the options given below :
correct answer:- 2
Question 38
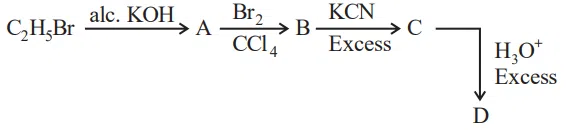
Acid D formed in above reaction is:
correct answer:- 2
Question 39
Which of the following has highly acidic hydrogen?
correct answer:- 4
Question 40
The molecular formula of second homologue in the homologous series of mono carboxylic acids is :
correct answer:- 2
Question 41
The products A and B formed in the following reaction scheme are respectively:
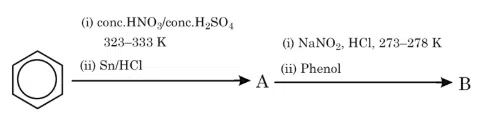
correct answer:- 3
Question 42
The metal that shows highest and maximum number of oxidation state is :
correct answer:- 2
Question 43
The spin-only magnetic moment value of the ion among $$Ti^{2+}, V^{2+}, Co^{3+}$$ and $$Cr^{2+}$$, that acts as strong oxidising agent in aqueous solution is ______ BM (Near integer). (Given atomic numbers : $$Ti : 22, V : 23, Cr : 24, Co : 27$$)
correct answer:- 5
Question 44
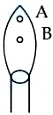
In a borax bead test under hot condition, a metal salt (one from the given) is heated at point B of the flame, resulted in green colour salt bead. The spin-only magnetic moment value of the salt is ______ BM (Nearest integer) [Given atomic number of $$Cu = 29, Ni = 28, Mn = 25, Fe = 26$$]
correct answer:- 6
Question 45
The number of elements from the following that do not belong to lanthanoids is $$Eu, Cm, Er, Tb, Yb$$ and $$Lu$$
correct answer:- 3
Question 46
Among $$CrO$$, $$Cr_2O_3$$ and $$CrO_3$$, the sum of spin-only magnetic moment values of basic and amphoteric oxides is _______ $$\times 10^{-2}$$ BM (nearest integer). (Given atomic number of Cr is 24)
correct answer:- 877
Question 47
The difference in the 'spin-only' magnetic moment values of $$KMnO_4$$ and the manganese product formed during titration of $$KMnO_4$$ against oxalic acid in acidic medium is _______ BM. (nearest integer)
correct answer:- 6
Question 48
Arrange the following elements in the increasing order of number of unpaired electrons in it. (A) Sc (B) Cr (C) V (D) Ti (E) Mn. Choose the correct answer from the options given below :
correct answer:- 3
Question 49
Among $$VO_2^{+}$$, $$MnO_4^{-}$$ and $$Cr_2O_7^{2-}$$, the spin-only magnetic moment value of the species with least oxidising ability is ___________ BM (Nearest integer). (Given atomic number $$V = 23, Mn = 25, Cr = 24$$)
correct answer:- 0
Question 50
The 'spin only' magnetic moment value of $$MO_4^{2-}$$ is ______ BM. (Where M is a metal having least metallic radii among $$Sc, Ti, V, Cr, Mn$$ and $$Zn$$). (Given atomic number: $$Sc = 21, Ti = 22, V = 23, Cr = 24, Mn = 25$$ and $$Zn = 30$$)
correct answer:- 0

 Group
Group






.webp)















