Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The compounds which give positive Fehling's test are :
(A).
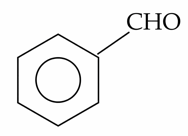
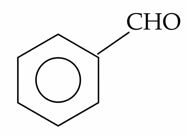
(B).
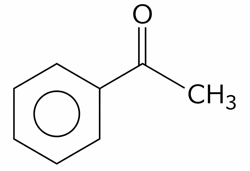
(C). $$ HOCH_{2} - CO - (CHOH)_{3}-CH_{2}-OH $$
(D). $$CH_{3}-CHO$$
(E).
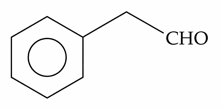
Choose the correct answer from the options given below :
A) Benzaldehyde: Gives a negative result. Although it is an aldehyde, aromatic aldehydes are generally not oxidized by the relatively weak Fehling's solution.
B) Acetophenone: Gives a negative result. It is a ketone, and ketones lack the oxidizable hydrogen atom attached to the carbonyl carbon necessary to reduce Fehling's solution.
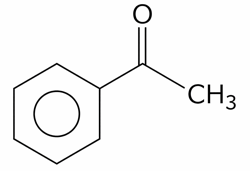
C) Fructose: Gives a positive result. Despite being a keto-hexose, it undergoes tautomerization under the alkaline conditions of the test to form oxidizable aldoses.
$$ HOCH_{2} - CO - (CHOH)_{3}-CH_{2}-OH $$
D) Acetaldehyde: Gives a positive result. As an aliphatic aldehyde, it is easily oxidized by the $$Cu^{2+}$$ ions in Fehling's solution to form a carboxylic acid and a red precipitate of $$Cu_2O$$.
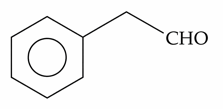
E) Phenylacetaldehyde: Gives a positive result. Even though it contains an aromatic ring, the aldehyde group is attached to an aliphatic side chain, allowing it to behave like a typical aliphatic aldehyde in this test.

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
