Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Match List - I with List - II.

choose the correct answer from the options given below :
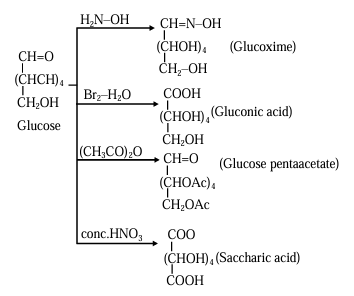
Glucose structure
C₁ = -CHO (aldehyde carbon)
C₂-C₅ = -CHOH
C₆ = -CH₂OH
A. Hydroxylamine
Reaction name: Oxime formation (nucleophilic addition to carbonyl)
What reacts:
C₁ (aldehyde carbon)
Change:
-CHO → -CH=NOH
Product: Glucoxime
B. Br₂ / H₂O
Reaction name: Mild oxidation of aldehyde
What reacts:
Only C₁ (aldehyde carbon)
Change:
-CHO → -COOH
Product: Gluconic acid
C. Excess acetic anhydride ((CH₃CO)₂O)
Reaction name: Acetylation (ester formation)
What reacts:
All hydroxyl groups: C₂, C₃, C₄, C₅, C₆
Change:
-OH → -OCOCH₃
Note:
C₁ remains as aldehyde
Product: Glucose pentaacetate
D. Concentrated HNO₃
Reaction name: Strong oxidation
What reacts:
C₁ (-CHO) and C₆ (-CH₂OH)
Change:
-CHO → -COOH
-CH₂OH → -COOH
Product: Saccharic acid
Final Matching
A - IV (Glucoxime)
B - I (Gluconic acid)
C - II (Glucose pentaacetate)
D - III (Saccharic acid)

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
