Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
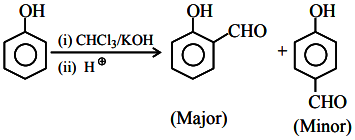
Given below are two statements:
Statement I: Phenol on treatment with . $$CHCL_{3}$$/aq. $$KOH$$ under refluxing condition, followed by acidification produces $$p$$-hydroxy benzaldehyde as the major product and $$o$$-hydroxy benzaldehyde as the minor product.
Statement II: The mixture of $$p$$-hydroxybenzaldehyde and $$o$$-
hydroxybenzaldehyde can be easily separated through steam distillation.
In the light of the above statements, choose the correct answer from the options given below

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
