Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
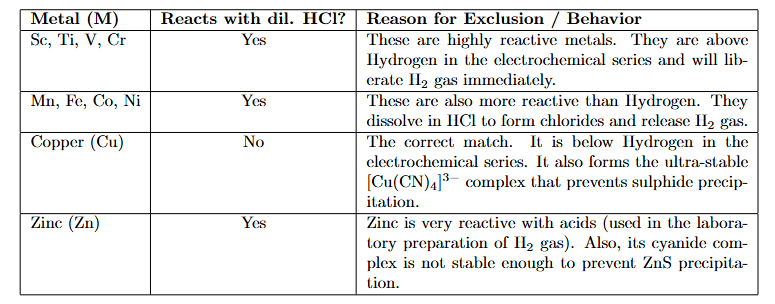
A first row transition metal (M) does not liberate $$H_{2}$$ gas from dilute HCI. 1 mol of aqueous solution of $$MSO_{4}$$ is treated with excess of aqueous KCN and then $$H_{2} S(g)$$ is passed through the solution. The amount of MS (metal sulphide) formed from the above reaction is _______ mol.

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
