Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Among the given transition metals, we need to find which shows the highest and maximum number of oxidation states.
Manganese (Mn): Electronic configuration [Ar] $$3d^5 4s^2$$. It shows oxidation states from +2 to +7 (total 6 different oxidation states). The maximum oxidation state is +7 (in $$KMnO_4$$), which equals the total number of 3d + 4s electrons (5 + 2 = 7).
Iron (Fe): [Ar] $$3d^6 4s^2$$. Shows up to +6 (rare). Common: +2, +3.
Cobalt (Co): [Ar] $$3d^7 4s^2$$. Shows up to +4. Common: +2, +3.
Titanium (Ti): [Ar] $$3d^2 4s^2$$. Shows up to +4.
Mn shows the highest maximum oxidation state (+7) and the maximum number of oxidation states among the 3d transition metals.
The correct answer is Option (2): Mn.
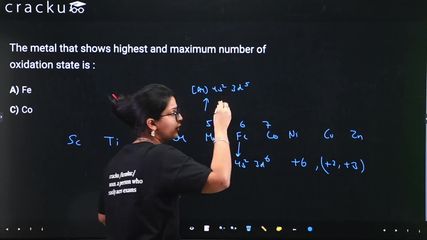
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
