Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Among $$VO_2^{+}$$, $$MnO_4^{-}$$ and $$Cr_2O_7^{2-}$$, the spin-only magnetic moment value of the species with least oxidising ability is ___________ BM (Nearest integer). (Given atomic number $$V = 23, Mn = 25, Cr = 24$$)
Correct Answer: 0
We need to find the spin-only magnetic moment of the species with the least oxidising ability among $$VO_2^+$$, $$MnO_4^-$$, and $$Cr_2O_7^{2-}$$.
Oxidising ability is related to the ease with which the species can be reduced. The higher the reduction potential, the stronger the oxidising agent. Among common oxidising agents in acidic medium: $$MnO_4^- > Cr_2O_7^{2-} > VO_2^+$$ in terms of oxidising strength.
The species with the least oxidising ability is $$VO_2^+$$.
Find the oxidation state and d-electron configuration of V in $$VO_2^+$$:
Let the oxidation state of V be $$x$$. Then $$x + 2(-2) = +1$$, so $$x = +5$$.
Vanadium (Z = 23) has the configuration [Ar]3d$$^3$$4s$$^2$$. V$$^{5+}$$ loses all 5 valence electrons, giving [Ar] = 3d$$^0$$.
Calculate the spin-only magnetic moment using the formula:
$$\mu = \sqrt{n(n+2)} \, \text{BM}$$
where $$n$$ is the number of unpaired electrons. Since V$$^{5+}$$ has d$$^0$$ configuration, $$n = 0$$:
$$\mu = \sqrt{0(0+2)} = 0 \, \text{BM}$$
The answer is 0.
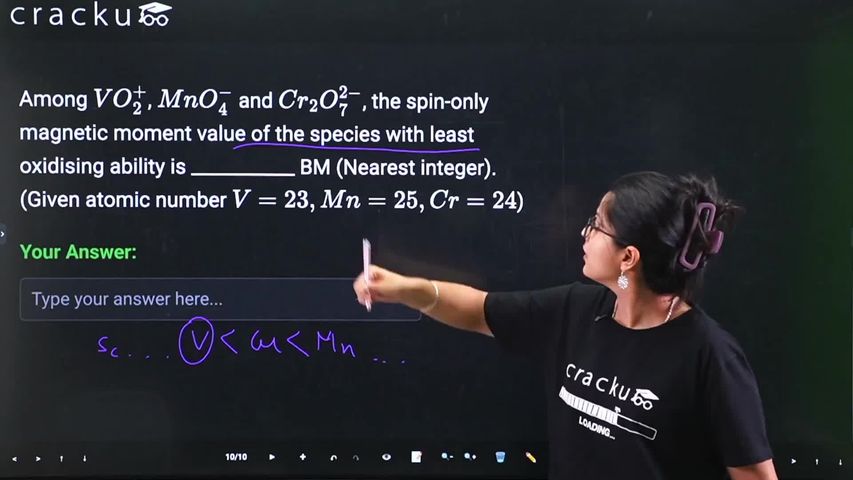
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
