Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
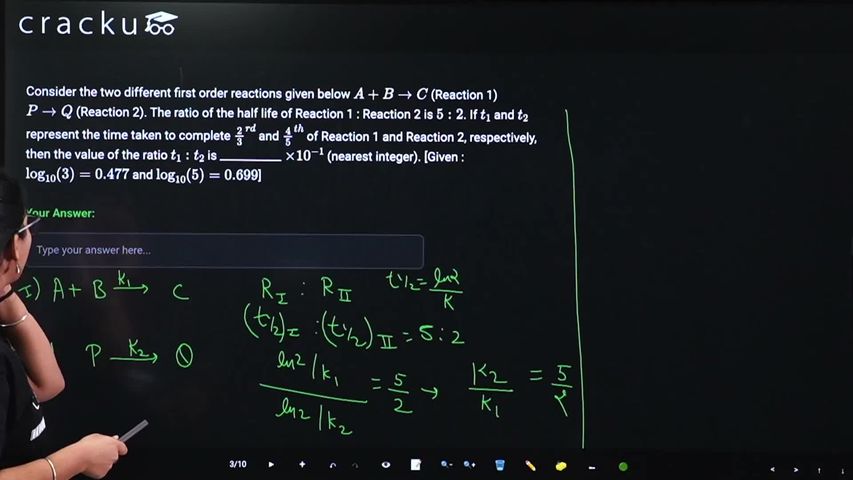
Consider the two different first order reactions given below $$A + B \rightarrow C$$ (Reaction 1) $$P \rightarrow Q$$ (Reaction 2). The ratio of the half life of Reaction 1 : Reaction 2 is $$5 : 2$$. If $$t_1$$ and $$t_2$$ represent the time taken to complete $$\frac{2}{3}^{rd}$$ and $$\frac{4}{5}^{th}$$ of Reaction 1 and Reaction 2, respectively, then the value of the ratio $$t_1 : t_2$$ is ___________ $$\times 10^{-1}$$ (nearest integer). [Given : $$\log_{10}(3) = 0.477$$ and $$\log_{10}(5) = 0.699$$]
Correct Answer: 17
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
