Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
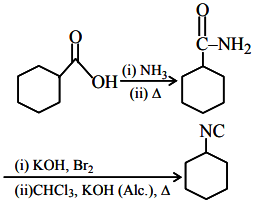
Given sequence uses:
NH₃/Δ → amide formation, Br₂/KOH → Hofmann bromamide degradation, CHCl₃/KOH → Carbylamine reaction (isocyanide formation)
A: Carboxylic acid (cyclohexyl-COOH)
NH₃/Δ → cyclohexyl-CONH₂ (amide formation)
Br₂/KOH → cyclohexyl-NH₂ (Hofmann degradation, one C less)
CHCl₃/KOH → cyclohexyl-NC (Carbylamine)
Gives required product → correct
B: Aldehyde (cyclohexyl-CH₂-CHO)
NH₃ does not convert aldehyde to amide under these conditions
No proper substrate for Hofmann reaction
Fails
C: Ketone (cyclohexyl-CO-CH₃)
NH₃ forms imine, not amide
Hofmann reaction needs amide → not possible
Fails
D: Amide (cyclohexyl-CONH₂)
Already amide, so NH₃/Δ does nothing
Br₂/KOH → cyclohexyl-NH₂
CHCl₃/KOH → cyclohexyl-NC
But initial step is unnecessary and does not match sequence properly
Not correct as per given pathway
Final answer: A

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
